The Research Whisperer
Just like the thesis whisperer – but with more money, how to make a simple research budget.

Every research project needs a budget*.
If you are applying for funding, you must say what you are planning to spend that funding on. More than that, you need to show how spending that money will help you to answer your research question .
So, developing the budget is the perfect time to plan your project clearly . A good budget shows the assessors that you have thought about your research in detail and, if it is done well, it can serve as a great, convincing overview of the project.
Here are five steps to create a simple budget for your research project.

1. List your activities
Make a list of everything that you plan to do in the project, and who is going to do it.
Take your methodology and turn it into a step-by-step plan. Have you said that you will interview 50 people? Write it on your list.
Are you performing statistical analysis on your sample? Write it down.
Think through the implications of what you are going to do. Do you need to use a Thingatron? Note down that you will need to buy it, install it, and commission it.
What about travel? Write down each trip separately. Be specific. You can’t just go to ‘South East Asia’ to do fieldwork. You need to go to Kuala Lumpur to interview X number of people over Y weeks, then the same again for Singapore and Jakarta.
Your budget list might look like this:
- I’m going to do 10 interviews in Kuala Lumpur; 10 interviews in Singapore; 10 interviews in Jakarta by me.
- I’ll need teaching release for three months for fieldwork.
- I’ll need Flights to KL, Singapore, Jakarta and back to Melbourne.
- I’ll need Accommodation for a month in each place, plus per diem.
- The transcription service will transcribe the 30 interviews.
- I’ll analysis the transcribed results. (No teaching release required – I’ll do it in my meagre research time allowance.)
- I’ll need a Thingatron X32C to do the trials.
- Thing Inc will need to install the Thingatron. (I wonder how long that will take.)
- The research assistant will do three trials a month with the Thingatron.
- I’ll need to hire a research assistant (1 day per week for a year at Level B1.)
- The research assistant will do the statistical analysis of the Thingatron results.
- I’ll do the writing up in my research allowance time.
By the end, you should feel like you have thought through the entire project in detail. You should be able to walk someone else through the project, so grab a critical friend and read the list to them. If they ask questions, write down the answers.
This will help you to get to the level of specificity you need for the next step.
2. Check the rules again
You’ve already read the funding rules, right? If not, go and read them now – I’ll wait right here until you get back.
Once you’ve listed everything you want to do, go back and read the specific rules for budgets again. What is and isn’t allowed? The funding scheme won’t pay for equipment – you’ll need to fund your Thingatron from somewhere else. Cross it off.
Some schemes won’t fund people. Others won’t fund travel. It is important to know what you need for your project. It is just as important to know what you can include in the application that you are writing right now.
Most funding schemes won’t fund infrastructure (like building costs) and other things that aren’t directly related to the project. Some will, though. If they do, you should include overheads (i.e. the general costs that your organisation needs to keep running). This includes the cost of basics like power and lighting; desks and chairs; and cleaners and security staff. It also includes service areas like the university library. Ask your finance officer for help with this. Often, it is a percentage of the overall cost of the project.
If you are hiring people, don’t forget to use the right salary rate and include salary on-costs. These are the extra costs that an organisation has to pay for an employee, but that doesn’t appear in their pay check. This might include things like superannuation, leave loading, insurance, and payroll tax. Once again, your finance officer can help with this.
Your budget list might now look like this:
- 10 interviews in Kuala Lumpur; 10 interviews in Singapore; 10 interviews in Jakarta by me.
- Teaching release for three months for fieldwork.
- Flights to KL, Singapore, Jakarta and back to Melbourne.
- Accommodation for a month in each place, plus per diem, plus travel insurance (rule 3F).
- Transcription of 30 interviews, by the transcription service.
- Analysis of transcribed results, by me. No teaching release required.
- Purchase and install Thingatron X32C, by Thing Inc . Not allowed by rule 3C . Organise access to Thingatron via partner organistion – this is an in-kind contribution to the project.
- Three trials a month with Thingatron, by research assistant.
- Statistical analysis of Thingatron results, by research assistant.
- Research assistant: 1 day per week for a year at Level B1, plus 25.91% salary on-costs.
- Overheads at 125% of total cash request, as per rule 3H.
3. Cost each item
For each item on your list, find a reasonable cost for it . Are you going to interview the fifty people and do the statistical analysis yourself? If so, do you need time release from teaching? How much time? What is your salary for that period of time, or how much will it cost to hire a replacement? Don’t forget any hidden costs, like salary on-costs.
If you aren’t going to do the work yourself, work out how long you need a research assistant for. Be realistic. Work out what level you want to employ them at, and find out how much that costs.
How much is your Thingatron going to cost? Sometimes, you can just look that stuff up on the web. Other times, you’ll need to ring a supplier, particularly if there are delivery and installation costs.
Jump on a travel website and find reasonable costs for travel to Kuala Lumpur and the other places. Find accommodation costs for the period that you are planning to stay, and work out living expenses. Your university, or your government, may have per diem rates for travel like this.
Make a note of where you got each of your estimates from. This will be handy later, when you write the budget justification.
- 10 interviews in Kuala Lumpur; 10 interviews in Singapore; 10 interviews in Jakarta by me (see below for travel costs).
- Teaching release for three months for fieldwork = $25,342 – advice from finance officer.
- Flights to KL ($775), Singapore ($564), Jakarta ($726), Melbourne ($535) – Blue Sky airlines, return economy.
- Accommodation for a month in each place (KL: $3,500; Sing: $4,245; Jak: $2,750 – long stay, three star accommodation as per TripAdviser).
- Per diem for three months (60 days x $125 per day – University travel rules).
- Travel insurance (rule 3F): $145 – University travel insurance calculator .
- Transcription of 30 interviews, by the transcription service: 30 interviews x 60 minutes per interview x $2.75 per minute – Quote from transcription service, accented voices rate.
- Analysis of transcribed results, by me. No teaching release required. (In-kind contribution of university worth $2,112 for one week of my time – advice from finance officer ).
- Purchase and install Thingatron X32C, by Thing Inc . Not allowed by rule 3C. Organise access to Thingatron via partner organistion – this is an in-kind contribution to the project. ($2,435 in-kind – quote from partner organisation, at ‘favoured client’ rate.)
- Research assistant: 1 day per week for a year at Level B1, plus 25.91% salary on-costs. $12,456 – advice from finance officer.
Things are getting messy, but the next step will tidy it up.
4. Put it in a spreadsheet
Some people work naturally in spreadsheets (like Excel). Others don’t. If you don’t like Excel, tough. You are going to be doing research budgets for the rest of your research life.
When you are working with budgets, a spreadsheet is the right tool for the job, so learn to use it! Learn enough to construct a simple budget – adding things up and multiplying things together will get you through most of it. Go and do a course if you have to.
For a start, your spreadsheet will multiply things like 7 days in Kuala Lumpur at $89.52 per day, and it will also add up all of your sub-totals for you.
If your budget doesn’t add up properly (because, for example, you constructed it as a table in Word), two things will happen. First, you will look foolish. Secondly, and more importantly, people will lose confidence in all your other numbers, too. If your total is wrong, they will start to question the validity of the rest of your budget. You don’t want that.
If you are shy of maths, then Excel is your friend. It will do most of the heavy lifting for you.
For this exercise, the trick is to put each number on a new line. Here is how it might look.
| Budget items | Number of items | Cost per item | Total cash cost | In-kind cost | Notes |
|---|---|---|---|---|---|
| Melbourne – Kuala Lumpur economy airfare | 1 | $775.00 | $775.00 | Blue Sky Airlines | |
| 1 month accommodation | 1 | $3,500.00 | $3,500.00 | 1 month x long stay via TripAdvisor | |
| 30 days per diem | 30 | $125.00 | $3,750.00 | University travel rules | |
| Kuala Lumpur – Singapore economy airfare | 1 | $564.00 | $564.00 | Blue Sky Airlines | |
| 1 month accommodation | 1 | $4,245.00 | $4,245.00 | 1 month x long stay via TripAdvisor | |
| 30 days per diem | 30 | $125.00 | $3,750.00 | University travel rules | |
| Singapore – Jakarta economy airfare | 1 | $726.00 | $726.00 | Blue Sky Airlines | |
| 1 month accommodation | 1 | $2,750.00 | $2,750.00 | 1 month x long stay via TripAdvisor | |
| 30 days per diem | 30 | $125.00 | $3,750.00 | University travel rules | |
| Jakarta – Melbourne economy airfare | 1 | $535.00 | $535.00 | Blue Sky Airlines | |
| Travel insurance: 90 days, South East Asia | 90 | $1.61 | $145.00 | University travel rules | |
| Transcription: 30 interviews with foreign accents | 1800 | $2.75 | $4,950.00 | Quote from transcription service | |
| Access to Thingatron | $2,435.00 | Favoured client rate, Thing Inc. | |||
| Chief Investigator: 0.2 of Academic D.2 | $36,457.00 | Includes 25.91% salary on-costs | |||
| Teaching relief: 90 days of Academic D.2 | $25,342.00 | Includes 25.91% salary on-costs | |||
| Research Assistant: 0.1 of Academic B.1 | $12,456.00 | Includes 25.91% salary on-costs | |||
| Sub-total | |||||
| Overheads | $84,047.50 | University overheads at 125% | |||
| Total |
5. Justify it
Accompanying every budget is a budget justification. For each item in your budget, you need to answer two questions:
- Why do you need this money?
- Where did you get your figures from?
The budget justification links your budget to your project plan and back again. Everything item in your budget should be listed in your budget justification, so take the list from your budget and paste it into your budget justification.
For each item, give a short paragraph that says why you need it. Refer back to the project plan and expand on what is there. For example, if you have listed a research assistant in your application, this is a perfect opportunity to say what the research assistant will be doing.
Also, for each item, show where you got your figures from. For a research assistant, this might mean talking about the level of responsibility required, so people can understand why you chose the salary level. For a flight, it might be as easy as saying: “Blue Sky airlines economy return flight.”
Here is an example for just one aspect of the budget:
Fieldwork: Kuala Lumpur
Past experience has shown that one month allows enough time to refine and localise interview questions with research partners at University of Malaya, test interview instrument, recruit participants, conduct ten x one-hour interviews with field notes. In addition, the novel methodology will be presented at CONF2015, to be held in Malaysia in February 2015.
Melbourne – Kuala Lumpur economy airfare is based on current Blue Sky Airlines rates. Note that airfares have been kept to a minimum by travelling from country to country, rather than returning to Australia.
1 month accommodation is based on three star, long stay accommodation rates provided by TripAdvisor.
30 days per diem rate is based on standard university rates for South-East Asia.
Pro tip: Use the same nomenclature everywhere. If you list a Thingatron X32C in your budget, then call it a Thingatron X32C in your budget justification and project plan. In an ideal world, someone should be able to flip from the project plan, to the budget and to the budget justification and back again and always know exactly where they are.
- Project plan: “Doing fieldwork in Malaysia? Whereabouts?” Flips to budget.
- Budget: “A month in Kuala Lumpur – OK. Why a month?” Flips to budget justification.
- Budget justification: “Ah, the field work happens at the same time as the conference. Now I get it. So, what are they presenting at the conference?” Flips back to the project description…
So, there you have it: Make a list; check the rules; cost everything; spreadsheet it; and then justify it. Budget done. Good job, team!
This article builds on several previous articles. I have shamelessly stolen from them.
- Constructing your budget – Jonathan O’Donnell.
- What makes a winning budget ? – Jonathan O’Donnell.
- How NOT to pad your budget – Tseen Khoo.
- Conquer the budget, conquer the project – Tseen Khoo.
- Research on a shoestring – Emily Kothe.
- How to make a simple Gantt chart – Jonathan O’Donnell.
* Actually, there are some grant schemes that give you a fixed amount of money, which I think is a really great idea . However, you will still need to work out what you are going to spend the money on, so you will still need a budget at some stage, even if you don’t need it for the application.
Also in the ‘simple grant’ series:
- How to write a simple research methods section .
- How to make a simple Gantt chart .
Share this:
28 comments.
This has saved my day!
Happy to help, Malba.
Like Liked by 1 person
[…] you be putting in a bid for funding? Are there costs involved, such as travel or equipment costs? Research Whisperer’s post on research budgets may help you […]
I’ve posted a link to this article of Jonathan’s in the Australasian Research Management Society LinkedIn group as well, as I’m sure lots of other people will want to share this.
Thanks, Miriam.
This is great! Humorous way to talk explain a serious subject and could be helpful in designing budgets for outreach grants, as well. Thanks!
Thanks, Jackie
If you are interested, I have another one on how to do a timeline: https://theresearchwhisperer.wordpress.com/2011/09/13/gantt-chart/
[…] really useful information regarding budget development can be found on the Research Whisperer Blog here. Any other thoughts and suggestions are welcome – what are your tips to developing a good […]
[…] it gets you to the level of specificity that you need for a detailed methods section. Similarly, working out a budget for your workshops will force you to be specific about how many people will be attending (venue […]
A friend of mine recently commented by e-mail:
I was interested in your blog “How to make a simple research budget”, particularly the statement: “Think through the implications of what you are going to do. Do you need to use a Thingatron? Note down that you will need to buy it, install it, and commission it.”
From my limited experience so far, I’d think you could add:
“Who else is nearby who might share the costs of the Thingatron? If it’s a big capital outlay, and you’re only going to use it to 34% of it’s capacity, sharing can make the new purchase much easier to justify. But how will this fit into your grant? And then it’s got to be maintained – the little old chap who used to just do all that odd mix of electrickery and persuasion to every machine in the lab got retrenched in the last round. You can run it into the ground. But that means you won’t have a reliable, stable Thingatron all ready to run when you apply for the follow-on grant in two years.”
[…] (For more on this process, take a look at How to Write a Simple Project Budget.) […]
[…] Source: How to make a simple research budget […]
This is such a big help! Thank You!
No worries, Claudine. Happy to help.
Would you like to share the link of the article which was wrote about funding rules? I can’t find it. Many thanks!
Hello there – do you mean this post? https://theresearchwhisperer.wordpress.com/2012/02/14/reading-guidelines
Thank @tseen khoo, very useful tips. I also want to understand more about 3C 3F 3H. What do they stand for? Can you help me find out which posts talk about that. Thank again.
[…] mount up rapidly, even if you are in a remote and developing part of the world. Putting together a half decent budget early on and being aware of funding opportunities can help to avoid financial disaster half way […]
This is so amazing, it really helpful and educative. Happy unread this last week before my proposal was drafted.
Happy to help, Babayomi. Glad you liked it.
really useful! thanks kate
[…] “How to Make a Simple Research Budget,” by Jonathan O’Donnell on The Research Whisperer […]
[…] offering services that ran pretty expensive. until I found this one. It guided me through making a simple budget. The information feels sort of like a university graduate research paper but having analysed […]
[…] Advice on writing research proposals for industry […]
[…] research serves as the bedrock of informed budgeting. Explore the average costs of accommodation, transportation, meals, and activities in your chosen […]
Leave a comment Cancel reply
This site uses Akismet to reduce spam. Learn how your comment data is processed .
- Already have a WordPress.com account? Log in now.
- Subscribe Subscribed
- Copy shortlink
- Report this content
- View post in Reader
- Manage subscriptions
- Collapse this bar
Designing a Research Proposal in Qualitative Research
- First Online: 27 October 2022
Cite this chapter

- Md. Ismail Hossain 4 ,
- Nafiul Mehedi 4 &
- Iftakhar Ahmad 4
2687 Accesses
The chapter discusses designing a research proposal in qualitative research. The main objective is to outline the major components of a qualitative research proposal with example(s) so that the students and novice scholars easily get an understanding of a qualitative proposal. The chapter highlights the major components of a qualitative research proposal and discusses the steps involved in designing a proposal. In each step, an example is given with some essential tips. Following these steps and tips, a novice researcher can easily prepare a qualitative research proposal. Readers, especially undergraduate and master’s students, might use this as a guideline while preparing a thesis proposal. After reading this chapter, they can easily prepare a qualitative proposal.
This is a preview of subscription content, log in via an institution to check access.
Access this chapter
- Available as PDF
- Read on any device
- Instant download
- Own it forever
- Available as EPUB and PDF
- Compact, lightweight edition
- Dispatched in 3 to 5 business days
- Free shipping worldwide - see info
- Durable hardcover edition
Tax calculation will be finalised at checkout
Purchases are for personal use only
Institutional subscriptions
Abdulai, R. T., & Owusu-Ansah, A. (2014). Essential ingredients of a good research proposal for undergraduate and postgraduate students in the social sciences. SAGE Open, 4 (3), 2158244014548178.
Article Google Scholar
Ahmad, S., Wasim, S., Irfan, S., Gogoi, S., Srivastava, A., & Farheen, Z. (2019). Qualitative versus quantitative research. Population, 1 , 2.
Google Scholar
Al-Riyami, A. (2008). How to prepare a research proposal. Oman Medical Journal, 23 (2), 66.
Aspers, P., & Corte, U. (2019). What is qualitative in qualitative research? Qualitative Sociology, 42 (2), 139–160.
Balakumar, P., Inamdar, M. N., & Jagadeesh, G. (2013). The critical steps for successful research: The research proposal and scientific writing (A report on the pre-conference workshop held in conjunction with the 64th annual conference of the Indian Pharmaceutical Congress-2012). Journal of Pharmacology & Pharmacotherapeutics, 4 (2), 130.
Becker, H. (1996). The epistemology of qualitative research. In R. Jessor, A. Colby & R. A Shweder (Eds.), Ethnography and human development: Context and meaning in social inquiry .
Boeije, H. (2010). Analysis in qualitative research . Los Angeles Sage Publications.
Bryman, A., Bresnen, M., Beardsworth, A., & Keil, T. (1988). Qualitative research and the study of leadership. Human Relations, 41 (1), 13–29.
Campbell, D. T., & Stanley, J. C. (2015). Experimental and quasi-experimental designs for research . Ravenio Books.
Creswell, J. W. (1994). Research design: Qualitative and quantitative approach . London: Publications.
Creswell, J. W. (2013). Research design: Qualitative, quantitative, and mixed methods approaches (4th Edn.). London: SAGE Publications, Inc.
Davis, B. (2021). What is the scope of the study in research proposal? Retrieved from https://www.mvorganizing.org/what-is-the-scope-of-the-study-in-research-proposal-4/#What_are_strengths_and_limitations . Accessed on August 28, 2021.
Denzin, N. K., & Lincoln, Y. S. (2005). Introduction: The discipline and practice of qualitative research. In N. K. Denzin & Y. S. Lincoln (Eds.), The Sage handbook of qualitative research (pp. 1–32). Sage Publications Ltd.
DJS Research. (2021). Qualitative research design . Retrieved from https://www.djsresearch.co.uk/glossary/item/Qualitative-Research-Design . Accessed on September 12, 2021.
Durrheim, K. (2006). Research design. In M. T. Blanche, M. J. T. Blanche, K. Durrheim, & D. Painter (Eds.), Research in practice: Applied methods for the social sciences (Vol. 2, pp. 33–59). Juta and Company Ltd.
Editage Insights. (2019). How do I present the scope of my study? Retrieved from https://www.editage.com/insights/how-do-i-present-scope-of-my-study . Accessed on August 31, 2021.
Fry, J., Scammell, J., & Barker, S. (2017). Drowning in muddied waters or swimming downstream? A critical analysis of literature reviewing in a phenomenological study through an exploration of the lifeworld, reflexivity and role of the researcher. Indo-Pacific Journal of Phenomenology , 17 (1).
Grove, S. K., Burns, N., & Gray, J. (2012). The practice of nursing research: Appraisal, synthesis, and generation of evidence . Elsevier Health Sciences.
Islam, M. R. (2019). Designing a Ph.D. proposal in qualitative research. In Social research methodology and new techniques in analysis, interpretation, and writing (pp. 1–22). IGI Global.
James, N., & Busher, H. (2009). Epistemological dimensions in qualitative research: The construction of knowledge online. SAGE Internet Research Methods , 5–18.
Liamputtong, P., & Ezzy, D. (2005). Qualitative research methods. Second . Oxford University Press.
Morse, J. M., & Field, P. A. (1996). The purpose of qualitative research. In Nursing research (pp. 1–17). Springer.
Mouton, J., & Marais, H. C. (1990). Basic concepts in the methodology of the social sciences (Revised). Human Sciences Research Council.
Parahoo, K. (2014). Nursing research: principles, process and issues (3rd ed.). Palgrave.
Pathak, V., Jena, B., & Kalra, S. (2013). Qualitative research. Perspectives in Clinical Research, 4 (3), 192. https://doi.org/10.4103/2229-3485.115389
Patton, A. J. (2001). Modelling time-varying exchange rate dependence using the conditional copula.
Pietilä, A. M., Nurmi, S. M., Halkoaho, A., & Kyngäs, H. (2020). Qualitative research: Ethical considerations. In The application of content analysis in nursing science research (pp. 49–69). Springer.
Rosenthal, M. (2016). Qualitative research methods: Why, when, and how to conduct interviews and focus groups in pharmacy research. Currents in Pharmacy Teaching and Learning, 8 (4), 509–516.
Russell, C. K., & Gregory, D. M. (2003). Evaluation of qualitative research studies. Evidence-Based Nursing, 6 (2), 36–40.
Sandelowski, M., & Barroso, J. (2003). Writing the proposal for a qualitative research methodology project. Qualitative Health Research, 13 (6), 781–820.
Strauss, A., & Corbin, J. (1990). Basics of qualitative research . Sage publications.
Walker, W. (2007). Ethical considerations in phenomenological research. Nurse researcher , 14 (3).
Wilson, A. (2015). A guide to phenomenological research. Nursing Standard, 29 (34), 38–43.
Download references
Author information
Authors and affiliations.
Department of Social Work, Shahjalal University of Science and Technology, Sylhet, Bangladesh
Md. Ismail Hossain, Nafiul Mehedi & Iftakhar Ahmad
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Md. Ismail Hossain .
Editor information
Editors and affiliations.
Centre for Family and Child Studies, Research Institute of Humanities and Social Sciences, University of Sharjah, Sharjah, United Arab Emirates
M. Rezaul Islam
Department of Development Studies, University of Dhaka, Dhaka, Bangladesh
Niaz Ahmed Khan
Department of Social Work, School of Humanities, University of Johannesburg, Johannesburg, South Africa
Rajendra Baikady
Rights and permissions
Reprints and permissions
Copyright information
© 2022 The Author(s), under exclusive license to Springer Nature Singapore Pte Ltd.
About this chapter
Hossain, M.I., Mehedi, N., Ahmad, I. (2022). Designing a Research Proposal in Qualitative Research. In: Islam, M.R., Khan, N.A., Baikady, R. (eds) Principles of Social Research Methodology. Springer, Singapore. https://doi.org/10.1007/978-981-19-5441-2_18
Download citation
DOI : https://doi.org/10.1007/978-981-19-5441-2_18
Published : 27 October 2022
Publisher Name : Springer, Singapore
Print ISBN : 978-981-19-5219-7
Online ISBN : 978-981-19-5441-2
eBook Packages : Social Sciences
Share this chapter
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Publish with us
Policies and ethics
- Find a journal
- Track your research
Let your curiosity lead the way:
Apply Today
- Arts & Sciences
- Graduate Studies in A&S
Creating a Budget
In general, while your research proposal outlines the academic significance of your study, the budget and budget narrative show that you have an understanding of what it will cost for you to be able to perform this research. Your proposed budget should identify all the expenses that are necessary and reasonable for the success of your project—no more and no less. The Office of Undergraduate Research understands that estimates, by definition, are imprecise, yet we encourage students applying for funding to research all aspects of their budgets with honest diligence.
If your research requires you to be in the field or in another city, state, or country, travel expenses may include transportation (airline, train, taxi, etc.), passport and visa fees, as well as fees for any vaccinations you may need to travel. Be sure to include anticipated major incidental expenses, such as printing, copying, fees for accessing archives, etc.
Please note that our funding restrictions prevent us from providing support for lab materials, equipment, software, hardware, etc.
Keep in mind these tips:
Convert all foreign currency figures to U.S. dollars.
Round all figures to whole dollars.
Make sure your budget and your proposal are consistent.
Identify areas where you are making efforts to save money!
Browse through these sample budgets for a better idea of how to outline your expenses and contact us if you have questions!
Sample Budget 1
Sample Budget 2
Sample Budget 3
Sample Budget 4
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- Indian J Anaesth
- v.60(9); 2016 Sep
How to write a research proposal?
Department of Anaesthesiology, Bangalore Medical College and Research Institute, Bengaluru, Karnataka, India
Devika Rani Duggappa
Writing the proposal of a research work in the present era is a challenging task due to the constantly evolving trends in the qualitative research design and the need to incorporate medical advances into the methodology. The proposal is a detailed plan or ‘blueprint’ for the intended study, and once it is completed, the research project should flow smoothly. Even today, many of the proposals at post-graduate evaluation committees and application proposals for funding are substandard. A search was conducted with keywords such as research proposal, writing proposal and qualitative using search engines, namely, PubMed and Google Scholar, and an attempt has been made to provide broad guidelines for writing a scientifically appropriate research proposal.
INTRODUCTION
A clean, well-thought-out proposal forms the backbone for the research itself and hence becomes the most important step in the process of conduct of research.[ 1 ] The objective of preparing a research proposal would be to obtain approvals from various committees including ethics committee [details under ‘Research methodology II’ section [ Table 1 ] in this issue of IJA) and to request for grants. However, there are very few universally accepted guidelines for preparation of a good quality research proposal. A search was performed with keywords such as research proposal, funding, qualitative and writing proposals using search engines, namely, PubMed, Google Scholar and Scopus.
Five ‘C’s while writing a literature review

BASIC REQUIREMENTS OF A RESEARCH PROPOSAL
A proposal needs to show how your work fits into what is already known about the topic and what new paradigm will it add to the literature, while specifying the question that the research will answer, establishing its significance, and the implications of the answer.[ 2 ] The proposal must be capable of convincing the evaluation committee about the credibility, achievability, practicality and reproducibility (repeatability) of the research design.[ 3 ] Four categories of audience with different expectations may be present in the evaluation committees, namely academic colleagues, policy-makers, practitioners and lay audiences who evaluate the research proposal. Tips for preparation of a good research proposal include; ‘be practical, be persuasive, make broader links, aim for crystal clarity and plan before you write’. A researcher must be balanced, with a realistic understanding of what can be achieved. Being persuasive implies that researcher must be able to convince other researchers, research funding agencies, educational institutions and supervisors that the research is worth getting approval. The aim of the researcher should be clearly stated in simple language that describes the research in a way that non-specialists can comprehend, without use of jargons. The proposal must not only demonstrate that it is based on an intelligent understanding of the existing literature but also show that the writer has thought about the time needed to conduct each stage of the research.[ 4 , 5 ]
CONTENTS OF A RESEARCH PROPOSAL
The contents or formats of a research proposal vary depending on the requirements of evaluation committee and are generally provided by the evaluation committee or the institution.
In general, a cover page should contain the (i) title of the proposal, (ii) name and affiliation of the researcher (principal investigator) and co-investigators, (iii) institutional affiliation (degree of the investigator and the name of institution where the study will be performed), details of contact such as phone numbers, E-mail id's and lines for signatures of investigators.
The main contents of the proposal may be presented under the following headings: (i) introduction, (ii) review of literature, (iii) aims and objectives, (iv) research design and methods, (v) ethical considerations, (vi) budget, (vii) appendices and (viii) citations.[ 4 ]
Introduction
It is also sometimes termed as ‘need for study’ or ‘abstract’. Introduction is an initial pitch of an idea; it sets the scene and puts the research in context.[ 6 ] The introduction should be designed to create interest in the reader about the topic and proposal. It should convey to the reader, what you want to do, what necessitates the study and your passion for the topic.[ 7 ] Some questions that can be used to assess the significance of the study are: (i) Who has an interest in the domain of inquiry? (ii) What do we already know about the topic? (iii) What has not been answered adequately in previous research and practice? (iv) How will this research add to knowledge, practice and policy in this area? Some of the evaluation committees, expect the last two questions, elaborated under a separate heading of ‘background and significance’.[ 8 ] Introduction should also contain the hypothesis behind the research design. If hypothesis cannot be constructed, the line of inquiry to be used in the research must be indicated.
Review of literature
It refers to all sources of scientific evidence pertaining to the topic in interest. In the present era of digitalisation and easy accessibility, there is an enormous amount of relevant data available, making it a challenge for the researcher to include all of it in his/her review.[ 9 ] It is crucial to structure this section intelligently so that the reader can grasp the argument related to your study in relation to that of other researchers, while still demonstrating to your readers that your work is original and innovative. It is preferable to summarise each article in a paragraph, highlighting the details pertinent to the topic of interest. The progression of review can move from the more general to the more focused studies, or a historical progression can be used to develop the story, without making it exhaustive.[ 1 ] Literature should include supporting data, disagreements and controversies. Five ‘C's may be kept in mind while writing a literature review[ 10 ] [ Table 1 ].
Aims and objectives
The research purpose (or goal or aim) gives a broad indication of what the researcher wishes to achieve in the research. The hypothesis to be tested can be the aim of the study. The objectives related to parameters or tools used to achieve the aim are generally categorised as primary and secondary objectives.
Research design and method
The objective here is to convince the reader that the overall research design and methods of analysis will correctly address the research problem and to impress upon the reader that the methodology/sources chosen are appropriate for the specific topic. It should be unmistakably tied to the specific aims of your study.
In this section, the methods and sources used to conduct the research must be discussed, including specific references to sites, databases, key texts or authors that will be indispensable to the project. There should be specific mention about the methodological approaches to be undertaken to gather information, about the techniques to be used to analyse it and about the tests of external validity to which researcher is committed.[ 10 , 11 ]
The components of this section include the following:[ 4 ]
Population and sample
Population refers to all the elements (individuals, objects or substances) that meet certain criteria for inclusion in a given universe,[ 12 ] and sample refers to subset of population which meets the inclusion criteria for enrolment into the study. The inclusion and exclusion criteria should be clearly defined. The details pertaining to sample size are discussed in the article “Sample size calculation: Basic priniciples” published in this issue of IJA.
Data collection
The researcher is expected to give a detailed account of the methodology adopted for collection of data, which include the time frame required for the research. The methodology should be tested for its validity and ensure that, in pursuit of achieving the results, the participant's life is not jeopardised. The author should anticipate and acknowledge any potential barrier and pitfall in carrying out the research design and explain plans to address them, thereby avoiding lacunae due to incomplete data collection. If the researcher is planning to acquire data through interviews or questionnaires, copy of the questions used for the same should be attached as an annexure with the proposal.
Rigor (soundness of the research)
This addresses the strength of the research with respect to its neutrality, consistency and applicability. Rigor must be reflected throughout the proposal.
It refers to the robustness of a research method against bias. The author should convey the measures taken to avoid bias, viz. blinding and randomisation, in an elaborate way, thus ensuring that the result obtained from the adopted method is purely as chance and not influenced by other confounding variables.
Consistency
Consistency considers whether the findings will be consistent if the inquiry was replicated with the same participants and in a similar context. This can be achieved by adopting standard and universally accepted methods and scales.
Applicability
Applicability refers to the degree to which the findings can be applied to different contexts and groups.[ 13 ]
Data analysis
This section deals with the reduction and reconstruction of data and its analysis including sample size calculation. The researcher is expected to explain the steps adopted for coding and sorting the data obtained. Various tests to be used to analyse the data for its robustness, significance should be clearly stated. Author should also mention the names of statistician and suitable software which will be used in due course of data analysis and their contribution to data analysis and sample calculation.[ 9 ]
Ethical considerations
Medical research introduces special moral and ethical problems that are not usually encountered by other researchers during data collection, and hence, the researcher should take special care in ensuring that ethical standards are met. Ethical considerations refer to the protection of the participants' rights (right to self-determination, right to privacy, right to autonomy and confidentiality, right to fair treatment and right to protection from discomfort and harm), obtaining informed consent and the institutional review process (ethical approval). The researcher needs to provide adequate information on each of these aspects.
Informed consent needs to be obtained from the participants (details discussed in further chapters), as well as the research site and the relevant authorities.
When the researcher prepares a research budget, he/she should predict and cost all aspects of the research and then add an additional allowance for unpredictable disasters, delays and rising costs. All items in the budget should be justified.
Appendices are documents that support the proposal and application. The appendices will be specific for each proposal but documents that are usually required include informed consent form, supporting documents, questionnaires, measurement tools and patient information of the study in layman's language.
As with any scholarly research paper, you must cite the sources you used in composing your proposal. Although the words ‘references and bibliography’ are different, they are used interchangeably. It refers to all references cited in the research proposal.
Successful, qualitative research proposals should communicate the researcher's knowledge of the field and method and convey the emergent nature of the qualitative design. The proposal should follow a discernible logic from the introduction to presentation of the appendices.
Financial support and sponsorship
Conflicts of interest.
There are no conflicts of interest.
Have a language expert improve your writing
Run a free plagiarism check in 10 minutes, generate accurate citations for free.
- Knowledge Base
- Starting the research process
- How to Write a Research Proposal | Examples & Templates
How to Write a Research Proposal | Examples & Templates
Published on October 12, 2022 by Shona McCombes and Tegan George. Revised on November 21, 2023.
A research proposal describes what you will investigate, why it’s important, and how you will conduct your research.
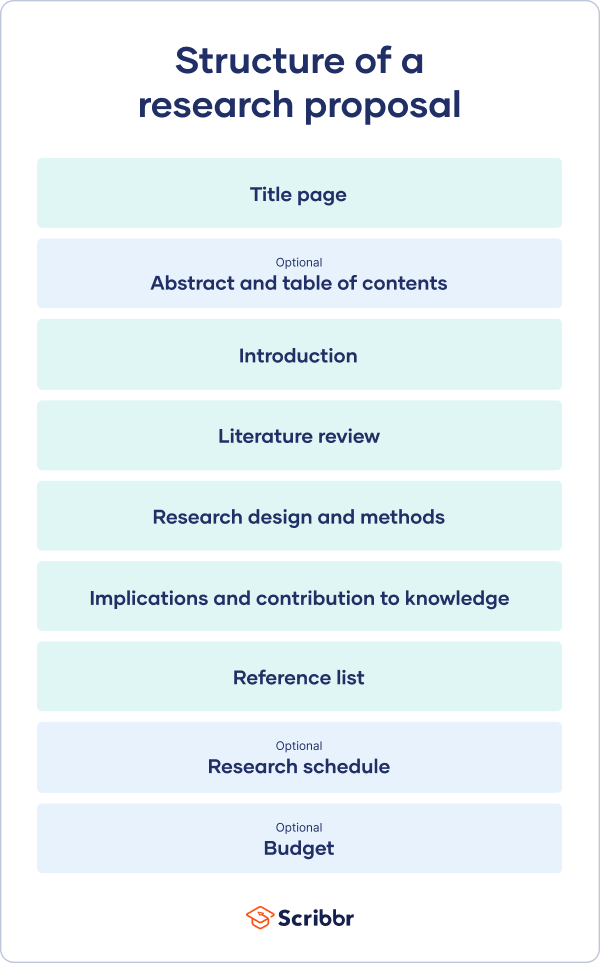
The format of a research proposal varies between fields, but most proposals will contain at least these elements:
Introduction
Literature review.
- Research design
Reference list
While the sections may vary, the overall objective is always the same. A research proposal serves as a blueprint and guide for your research plan, helping you get organized and feel confident in the path forward you choose to take.
Table of contents
Research proposal purpose, research proposal examples, research design and methods, contribution to knowledge, research schedule, other interesting articles, frequently asked questions about research proposals.
Academics often have to write research proposals to get funding for their projects. As a student, you might have to write a research proposal as part of a grad school application , or prior to starting your thesis or dissertation .
In addition to helping you figure out what your research can look like, a proposal can also serve to demonstrate why your project is worth pursuing to a funder, educational institution, or supervisor.
| Show your reader why your project is interesting, original, and important. | |
| Demonstrate your comfort and familiarity with your field. Show that you understand the current state of research on your topic. | |
| Make a case for your . Demonstrate that you have carefully thought about the data, tools, and procedures necessary to conduct your research. | |
| Confirm that your project is feasible within the timeline of your program or funding deadline. |
Research proposal length
The length of a research proposal can vary quite a bit. A bachelor’s or master’s thesis proposal can be just a few pages, while proposals for PhD dissertations or research funding are usually much longer and more detailed. Your supervisor can help you determine the best length for your work.
One trick to get started is to think of your proposal’s structure as a shorter version of your thesis or dissertation , only without the results , conclusion and discussion sections.
Download our research proposal template
Here's why students love Scribbr's proofreading services
Discover proofreading & editing
Writing a research proposal can be quite challenging, but a good starting point could be to look at some examples. We’ve included a few for you below.
- Example research proposal #1: “A Conceptual Framework for Scheduling Constraint Management”
- Example research proposal #2: “Medical Students as Mediators of Change in Tobacco Use”
Like your dissertation or thesis, the proposal will usually have a title page that includes:
- The proposed title of your project
- Your supervisor’s name
- Your institution and department
The first part of your proposal is the initial pitch for your project. Make sure it succinctly explains what you want to do and why.
Your introduction should:
- Introduce your topic
- Give necessary background and context
- Outline your problem statement and research questions
To guide your introduction , include information about:
- Who could have an interest in the topic (e.g., scientists, policymakers)
- How much is already known about the topic
- What is missing from this current knowledge
- What new insights your research will contribute
- Why you believe this research is worth doing
Prevent plagiarism. Run a free check.
As you get started, it’s important to demonstrate that you’re familiar with the most important research on your topic. A strong literature review shows your reader that your project has a solid foundation in existing knowledge or theory. It also shows that you’re not simply repeating what other people have already done or said, but rather using existing research as a jumping-off point for your own.
In this section, share exactly how your project will contribute to ongoing conversations in the field by:
- Comparing and contrasting the main theories, methods, and debates
- Examining the strengths and weaknesses of different approaches
- Explaining how will you build on, challenge, or synthesize prior scholarship
Following the literature review, restate your main objectives . This brings the focus back to your own project. Next, your research design or methodology section will describe your overall approach, and the practical steps you will take to answer your research questions.
| ? or ? , , or research design? | |
| , )? ? | |
| , , , )? | |
| ? |
To finish your proposal on a strong note, explore the potential implications of your research for your field. Emphasize again what you aim to contribute and why it matters.
For example, your results might have implications for:
- Improving best practices
- Informing policymaking decisions
- Strengthening a theory or model
- Challenging popular or scientific beliefs
- Creating a basis for future research
Last but not least, your research proposal must include correct citations for every source you have used, compiled in a reference list . To create citations quickly and easily, you can use our free APA citation generator .
Some institutions or funders require a detailed timeline of the project, asking you to forecast what you will do at each stage and how long it may take. While not always required, be sure to check the requirements of your project.
Here’s an example schedule to help you get started. You can also download a template at the button below.
Download our research schedule template
| Research phase | Objectives | Deadline |
|---|---|---|
| 1. Background research and literature review | 20th January | |
| 2. Research design planning | and data analysis methods | 13th February |
| 3. Data collection and preparation | with selected participants and code interviews | 24th March |
| 4. Data analysis | of interview transcripts | 22nd April |
| 5. Writing | 17th June | |
| 6. Revision | final work | 28th July |
If you are applying for research funding, chances are you will have to include a detailed budget. This shows your estimates of how much each part of your project will cost.
Make sure to check what type of costs the funding body will agree to cover. For each item, include:
- Cost : exactly how much money do you need?
- Justification : why is this cost necessary to complete the research?
- Source : how did you calculate the amount?
To determine your budget, think about:
- Travel costs : do you need to go somewhere to collect your data? How will you get there, and how much time will you need? What will you do there (e.g., interviews, archival research)?
- Materials : do you need access to any tools or technologies?
- Help : do you need to hire any research assistants for the project? What will they do, and how much will you pay them?
If you want to know more about the research process , methodology , research bias , or statistics , make sure to check out some of our other articles with explanations and examples.
Methodology
- Sampling methods
- Simple random sampling
- Stratified sampling
- Cluster sampling
- Likert scales
- Reproducibility
Statistics
- Null hypothesis
- Statistical power
- Probability distribution
- Effect size
- Poisson distribution
Research bias
- Optimism bias
- Cognitive bias
- Implicit bias
- Hawthorne effect
- Anchoring bias
- Explicit bias
Once you’ve decided on your research objectives , you need to explain them in your paper, at the end of your problem statement .
Keep your research objectives clear and concise, and use appropriate verbs to accurately convey the work that you will carry out for each one.
I will compare …
A research aim is a broad statement indicating the general purpose of your research project. It should appear in your introduction at the end of your problem statement , before your research objectives.
Research objectives are more specific than your research aim. They indicate the specific ways you’ll address the overarching aim.
A PhD, which is short for philosophiae doctor (doctor of philosophy in Latin), is the highest university degree that can be obtained. In a PhD, students spend 3–5 years writing a dissertation , which aims to make a significant, original contribution to current knowledge.
A PhD is intended to prepare students for a career as a researcher, whether that be in academia, the public sector, or the private sector.
A master’s is a 1- or 2-year graduate degree that can prepare you for a variety of careers.
All master’s involve graduate-level coursework. Some are research-intensive and intend to prepare students for further study in a PhD; these usually require their students to write a master’s thesis . Others focus on professional training for a specific career.
Critical thinking refers to the ability to evaluate information and to be aware of biases or assumptions, including your own.
Like information literacy , it involves evaluating arguments, identifying and solving problems in an objective and systematic way, and clearly communicating your ideas.
The best way to remember the difference between a research plan and a research proposal is that they have fundamentally different audiences. A research plan helps you, the researcher, organize your thoughts. On the other hand, a dissertation proposal or research proposal aims to convince others (e.g., a supervisor, a funding body, or a dissertation committee) that your research topic is relevant and worthy of being conducted.
Cite this Scribbr article
If you want to cite this source, you can copy and paste the citation or click the “Cite this Scribbr article” button to automatically add the citation to our free Citation Generator.
McCombes, S. & George, T. (2023, November 21). How to Write a Research Proposal | Examples & Templates. Scribbr. Retrieved June 18, 2024, from https://www.scribbr.com/research-process/research-proposal/
Is this article helpful?
Shona McCombes
Other students also liked, how to write a problem statement | guide & examples, writing strong research questions | criteria & examples, how to write a literature review | guide, examples, & templates, get unlimited documents corrected.
✔ Free APA citation check included ✔ Unlimited document corrections ✔ Specialized in correcting academic texts
- Develop a research budget
- Research Expertise Engine
- Precursors to research
- Funding Opportunities
- Grants vs contracts
- Sample Applications Library
- Factors to consider
- Internal Approval (formerly SFU Signature Sheet)
- Develop a research proposal
- Institutional support
- Review & submission
- Award & approval
- Award management
- Contracts & agreements
- Inventions & commercialization
- Ethics - human research
- Ethics - animal research
- Research safety
- Mobilizing Research
- Prizes & awards
- Training & events
- Forms & documents
On this page:
Basic components of a research budget, two models of budget development, other factors affecting your budget.
- Additional Resources
Budgets should provide the sponsor with an accurate assessment of all cost items and cost amounts that are deemed necessary and reasonable to carry out your project. They should be based upon your description or the statement of work. Budget justification provides more in-depth detail and reason for each cost and is often considered by reviewers as a good indicator of the feasibility of the research.
A research budget contains both direct costs and indirect costs (overhead), but the level of detail varies from sponsor to sponsor. The first step in developing a budget is to carefully read the guidelines of the funding opportunity being pursued.
There is no magic formula available for developing a budget but there are some basic steps to follow in order to develop an accurate budget:
- Define project tasks, timelines and milestones and determine the actual resources and costs required to complete these. Consider whether contingencies are needed (and confirm they are eligible expenses).
- Determine the eligible expense categories and maximum amount allowed by the sponsor. Adjust scope of the project to make sure proposed activities fit within the allowance.
- Categorize these costs (e.g., salaries, supplies, equipment…) per year, in some cases by quarter.
- Ensure that project scope and budget match. Include indirect costs of research as permitted by sponsor and the University policy.
The examples below developed by the University of British Columbia demonstrate two ways to include indirect costs in your budget.
- Price model: Indirect cost is built into each budget line item.
- Cost model: Indirect cost of research is presented as a separate line item.
Unless the sponsor specifies in writing that they require the indirect costs of research to be presented as a separate line item (Cost Model), the indirect cost should be built into each budget line item (Price Model). Indirect costs are normally included in the price of goods and services worldwide.
For example, you are developing a budget for a funding opportunity with an indirect cost rate of 25%. Your direct costs are $201,000 broken down by expense categories shown in the second column of the table below. The third and fourth colums present the two ways you can include the 25% overhead in your budget using the Price Model or the Cost Model, respectively:
| Line item description | Direct Cost | Price model (indirect cost built into each line item) | Cost model (indirect cost presented as a separate line item) |
|---|---|---|---|
| Salary: Post-Doctoral fellow * 1 | $42,000 | $52,500 | $42,000 |
| Salary: PhD student * 2 | $43,000 | $53,750 | $43,000 |
| Salary: Master's student * 3 | $54,000 | $67,500 | $54,000 |
| Digital devices | $26,000 | $32,500 | $26,000 |
| Consumables | $15,000 | $18,750 | $15,000 |
| Travel and subsistence | $21,000 | $26,250 | $21,000 |
| SFU Indirect Cost (25% of Direct Costs) | N/A | N/A | $50,250 |
In-kind and cash contributions, like other costs to the sponsored project, must be eligible and must be treated in a consistent and uniform manner in proposal preparation and in financial reporting.
Cash contributions
Cash contributions are actual cash transactions that can be documented in the accounting system. Examples of cash contributions include:
- allocation of compensated faculty and staff time to projects, or
- the purchasing of equipment by the university or other eligible sponsor for the benefit of the project.
In-kind contributions
In-kind contributions are both non-monetary or cash equivalent resources that can be given a cash value, such as goods and/or services in support of a research project or proposal. It is challenging to report on in-kind contribution, please make sure the numbers you use are well supported, consistent and easy to quantitate.
Examples of an in-kind contribution may include:
- Access to unique database or information
- Professional, analytical, and other donated services
- Employee salaries including benefits for time allocated to the project
- Study materials, technologies, or components
- Patents and licenses for use
- Use of facilities (e.g., lab or meeting spaces)
- Partner organization time spent participating in the project
- Eligible infrastructure items
Matching on sponsored projects
Some sponsored projects require the university and/or a third party to contribute a portion of the project costs–this contribution is known as matching.
Matching requirements may be in the form of an actual cash expenditure of funds or may be an “in-kind” match. For example:
- A 1:1 match would require $100 of a third-party matching for every $100 received from an agency.
- A 30% match would mean that of a total budget of $100, the agency would provide $70 and a third party would need to match $30.
Examples of agency programs that include some form of matching from a third party are:
- NSERC Collaborative Research and Development Grants
- NSERC Idea to Innovation Grants
- SSHRC Partnership Grants
- CIHR Industry Partnered Collaborative Research Program, and
- CIHR Proof of Principle Grants
Additional resources
- Current salary and benefit rates for graduate students and postdocs/research associates
- SFU Business and Travel Expense Policy
- Animal care services
- Vice-Chancellor
- Leadership and Governance
- Education Quality
- Sustainability
- Staff Directory
- Staff Profiles
- Staff Online
- Office of Human Resources
- Important Dates
- Accept and Enrol
- Student Forms
- Jobs for Students
- Future Students
- Scholarships
- Class Registration
- Online Courses
- Password Management
- Western Wifi - Wireless
- Accommodation
- The College
- Whitlam Institute
- Ask Western
- Staff Email
- WesternNow Staff Portal
- ResearchMaster
- Citrix Access
- Student Management System
- Exam Timetable
- Oracle Financials
- Casual Room Bookings
- Staff Profile Editor
- Vehicle Bookings
- Form Centre
- WSU SharePoint Portal
- Learning Guide Management System (LGMS)
- Student Email
- My Student Records (MySR)
- WesternLife
- WesternNow Student Portal
- My Exam Timetable
- Student Forms (eForms)
- Accept My Offer

Study with Us
- International
- Research Portal
- ResearchDirect
- Research Theme Program
- Researcher Development
- Funding Opportunities
- Preparing a Grant Application
- Research Ethics & Integrity
- Research Project Risk & Compliance
- Foreign Arrangements Scheme
- Managing Your Research Project
- Research Data Management
- Business Services
- Research Infrastructure
- Office of the DVC REI
- Research Services Update
- Contact Research Services
- Master of Research
- Research Degrees
- Find a Supervisor
- Graduate Research School
- Apply for a Research Degree
- Candidate Support and Resources
- HDR Knowledge Directory
- Research Ethics
- HDR Workshops
- Forms, Policies and Guidelines
- Giving to Western
- Bushfire and Natural Hazards
- Digital Health
- Future Food Systems
- RoZetta Institute
- Hawkesbury Institute for the Environment
- Ingham Institute
- Institute for Australian and Chinese Arts and Culture
- Institute for Culture and Society
- NICM Health Research Institute
- The MARCS Institute
- Translational Health Research Institute
- Australia India Water Centre
- Centre for Educational Research
- Centre for Infrastructure Engineering
- Centre for Research in Mathematics and Data Science
- Centre for Smart Modern Construction (c4SMC)
- Centre for Western Sydney
- Chinese Medicine Centre
- Global Centre for Land-Based Innovation
- International Centre for Neuromorphic Systems
- National Vegetable Protected Cropping Centre
- Transforming early Education And Child Health Research Centre (TeEACH)
- Urban Transformations Research Centre
- Writing and Society Research Centre
- Young and Resilient Research Centre
- Digital Humanities Research Group
- Humanitarian and Development Research Initiative (HADRI)
- Nanoscale Organisation and Dynamics Research Group
- Research at Western
- Research Impact
- - Research Portal
- - ResearchDirect
- - Researcher Development
- - Funding Opportunities
- - Definition of Research
- - Research or Consultancy Activity?
- - Writing a Project Description
- - Track Record Statement
- - Tips for ECRs
- - Developing a Budget
- - Budget Justification
- - Research Contracts
- - Research Codes
- - Research Project Risk and Compliance
- - Foreign Arrangements Scheme
- - Managing Your Project
- - Research Data Management
- Research Ethics and Integrity
- Research Management Solution (RMS)
- Research Participation Opportunities
Developing a Budget for Your Research Application
Budgets and budget justifications demonstrate feasibility, value for money and detail why you need an item for your project, as well as how you arrived at the costings.
Every research project has two budget categories: direct costs and indirect costs.
The University determines a set percentage for the indirect costs of funded research. Contact Grants Services for the correct figure to use.
Direct costs are costs integral to achieving the research objectives of a grant. The costs directly address the research objectives of the grant and relate to the research plan.
Direct cost examples:
- Personnel, e.g. research assistants, student stipends for PhDs, and staff costs. You need to factor in salary increases, on-costs (superannuation and payroll) and casual loadings . Always use the salary level and step corresponding with the skills and tasks required for the role. See the Position Descriptors in the relevant University Enterprise Agreement .
- Equipment, maintenance and travel (outline why you are going and for how long)
- Teaching relief
- Other (e.g. Consumables).
Indirect costs are institution costs that benefit and support research activities at the institution. Although they are necessary for the conduct of research and may be incurred during the project, they are costs that do not directly address the approved research objectives of a grant.
Indirect cost examples:
- Operations and maintenance of buildings (e.g. libraries, labs, meeting venues, IT such as computer access, specialist software, databases, secure cloud storage)
- Insurance, legal and financial services
- Hazardous waste disposal, and
- Regulatory and research compliance and administration of research services
All external research activities are expected to contribute to indirect costs except :
- Nationally competitive grants, such as ARC and NHMRC. This includes all Category 1 schemes.
- Registered charities listed on the ACNC register (opens in a new window)
- Grants transferred from another university
- Funding bodies that exclude or limit overheads or administrative costs (i.e. indirect costs) in their rules or guidelines
- Scholarships and internships
- Official Western Partnership projects
- Travel award type grants or facility usage type grants (e.g. Endeavour Fellowships, AINSE grants)
- Projects costed under $100,000 are discounted by waiving Western’s portion of the indirect costs.
Indirect costs are calculated by determining the direct costs first and then applying the indirect costs formula:
e.g. Direct costs = $50,000 x (indirect cost % figure) = Total project cost
Cash and in-kind support
Your project budget needs to include all cash and in-kind items it requires.
In-kind support is any non-cash contributions that a party gives to the project. In-kind can be contributed by Western Sydney University or by an external party, and can include:
- staff (e.g. time committed to the project which is not funded by the project)
- non-staff/infrastructure (e.g. if you are using lab space to conduct the project but are not receiving direct payment from the project to 'buy out' lab space)
- indirect costs

How to budget personnel and salaries
On-costs are direct costs associated with salary. These costs relate to superannuation, sick leave, payroll tax etc. and must be included your budget.
Access this link for more detail about Western on-costs
For the latest salary figures, please check with the Office of People
An example:
You are a Lead Chief Investigator (CI) on a non-Category 1 funding body project for one year. You commit 0.4 (FTE) of your time to the research = 2 days per week. You are paid at Academic Level E, Step 2, which is $188,944 per annum. You can calculate your salary inclusive of 28% on-costs as follows:
0.4 x 0.28 x 188,944 = 21,161.73
The budgeting of your salary, a direct cost of the research, should be listed as $21,161.73.
If your project covers three years, with the same or differing time commitments, you calculate this figure for each year of your project. Remember to factor in pay rises according to Step increases in multi-year grants.
You may also have a research assistant employed full-time for seven weeks at HEW Level 5, Step 3. You hire the assistant at the casual hourly rate of $48.97, which includes 25% leave loading. You add 16.5% on-costs to this figure:
48.97 x (35 x 7) = 11,997.65
11,997.65 x .165 = 1,979.50
1,979.50 + 11,997.65 = 13,977.15
The total cost to employ the research assistant is $13,977.15.
Note 1: the maximum period a person can be employed on a casual rate is 6 months.
Note 2: For some schemes, the funding provider stipulates a specific maximum rate for funding of salary on-costs, e.g. the Australian Research Council (ARC) funds on-costs at a rate of 30%, so you must use this figure.
- Grant Budget Calculator (Staff Login Required) (opens in a new window)
^ Back to top
Mobile options:
- Return to standard site
- Back to Top

International Students
Launch your career at UWS
- University Life
- Our Campuses
- Business and Community
- Undergraduate
- Postgraduate
- HDR Research
- Student Life
- Why Western
- The Academy
- Western Sydney University Online
- Misconduct Rule
- Study with Integrity
- Student Completions
- Student Support
- Services and Facilities
- Working with us
- Career Development
- Salary and Benefits
- Manager/Supervisor Toolkit
- Future Staff
- Staff Services
- Researchers
- Current Students
- Community and Industry
- Alumni Awards
- Alumni Spotlight
- Alumni Benefits
- Alumni Affinity Groups
- Alumni Publications
- Alumni Giving
Western Sydney University

- Emergency Help
- Right to Information
- Complaints Unit
- Accessibility
- Website Feedback
- Compliance Program
- Admissions Transparency
As the nation’s largest public research university, the Office of the Vice President for Research (OVPR) aims to catalyze, support and safeguard U-M research and scholarship activity.
The Office of the Vice President for Research oversees a variety of interdisciplinary units that collaborate with faculty, staff, students and external partners to catalyze, support and safeguard research and scholarship activity.
ORSP manages pre-award and some post-award research activity for U-M. We review contracts for sponsored projects applying regulatory, statutory and organizational knowledge to balance the university's mission, the sponsor's objectives, and the investigator's intellectual pursuits.
Ethics and compliance in research covers a broad range of activity from general guidelines about conducting research responsibly to specific regulations governing a type of research (e.g., human subjects research, export controls, conflict of interest).
eResearch is U-M's site for electronic research administration. Access: Regulatory Management (for IRB or IBC rDNA applications); Proposal Management (eRPM) for the e-routing, approval, and submission of proposals (PAFs) and Unfunded Agreements (UFAs) to external entities); and Animal Management (for IACUC protocols and ULAM).
Sponsored Programs manages the post-award financial activities of U-M's research enterprise and other sponsored activities to ensure compliance with applicable federal, state, and local laws as well as sponsor regulations. The Office of Contract Administration (OCA) is also part of the Office of Finance - Sponsored Programs.
- Develop Proposal Overview
- Blue Pages: Find an RA
- Cost Accounting Standards
- Budget Table Sample
- Direct vs. F&A Costs
- Direct Costs Other Than Salaries
- Facilities & Administrative Rates
- Full Recovery of F&A Costs - Calculating Space
- NRSA Stipend Levels and GSRA Max
- NIH Modular Budgets
- Participant Support Costs
- Salaries in Sponsored Projects
Staff Benefits
- Uniform Guidance Monitored Costs
- Clinical Trials
- Create Biosketch
- OVPR Cost Sharing
- About the University of Michigan
- NIH - Other Support Reporting
- NSF and Other Sponsors - Current and Pending Support
- Principal Investigator Definition
- Cost / Price Analysis
- Working with Subrecipients / Subrecipient Forms / Letter of Commitment
You are here
- Develop Proposal
Budget and Cost Resources
Quick links.
- Budget & Cost Resources
- UG Monitored Costs
- Direct and Indirect Costs
- Indirect Cost Rates
- Full Recovery of Indirect Costs - Calculating space for certain private sponsor
- Direct Costs (other than salaries)
- Staff Fringe Benefits
- GSRA Cost Estimates
The Principal Investigator has primary responsibility for budget planning, in consultation with the department chair or director of the research unit. Budgets for all sponsored proposals are subject to review by ORSP.
A Special Note About NIH Grant-Related Data Sharing Costs
Certain funding agencies (e.g. the National Institutes of Health (NIH)) are increasingly allowing data management and sharing (DMS) costs to be included as direct costs in proposal budgets.
NIH DMS costs should be shared in the appropriate cost category, e.g., personnel, equipment, supplies, and other expenses, following the instructions and providing details as instructed within the applicable form (e.g., R&R Budget Form or PHS 398 Modular Budget Form).
In most cases, the DMS Plan oversight at U-M will be provided by the principal investigator (PI) and other study personnel. If help is needed, consider these resources:
- OVPR Research Data Stewardship page - NIH DMSP resources
- U-M Library - Research Guides - Research Data Management
- U-M Navigate Webinar - NIH Data Management & Sharing Policy (Nov 1, 2022)
Budget Format
Sponsors often prescribe the budget format that must accompany the proposal, including the specific cost categories that should be identified. The format shown on the sample budget page may be used, however, if one is not specified by the sponsor. Be sure to check with your department, unit, school or college administrator to determine the best practice.
The budget should be subdivided into periods of 12-month duration (unless partial year funding is anticipated). A "starting date" should be specified, since it is essential to ensure accurate budget calculations. If cost-sharing is included, each budget period should include columns for both "Sponsor" and "University" costs. A budget summary should be included for proposals with multi-year funding. All budget entries should be rounded to the nearest whole dollar.
Salary and Wages
The salary category in the proposed budget should include the names and/or titles for all personnel involved in the project. The number of person months or percent effort to be applied to the project should also be shown. Total salary costs can be determined by applying the percentage of effort to the current salary rates. An appropriate escalation rate (e.g., 3%) should be used to determine salary requirements beyond the current fiscal year. While standard percentages are applied to make these calculations, no commitment and no constraint on the rate of increase for a given individual is implied by this procedure.
If a faculty member is working on several sponsored projects, care must be exercised to ensure that no more than 100 percent of effort is committed to the aggregate of all projects and other University responsibilities.
Summer salary:
Summer salary for faculty with academic year (AY) appointments can be figured at one-ninth of their institutional base salary for each month of summer effort. A maximum of two and one-half months may be included for the whole summer. Some sponsors, however, impose specific limitations on summer salaries. The National Science Foundation, for example, usually will not pay for more than two months of summer research at a rate of one-ninth of the AY salary per month.
Technical staff
Costs incurred for the same purpose in like circumstances must be treated consistently. For example, salaries of technical staff should be treated as direct costs wherever the work to be undertaken can be identified with a particular sponsored project. Direct charging of these costs may be accomplished by specifying individual positions within the project budget or through the use of recharge rates or specialized service facilities, as appropriate under the circumstances.
Administrative and clerical support
The salaries of administrative and clerical support staff normally should be treated as indirect costs . However, it may be appropriate to charge these costs directly to a sponsored project when the participation of the administrative/clerical staff being charged to a federal project meet all four of the following conditions as set forth in §200.413 of the Uniform Guidance:
“(1) Administrative or clerical services are integral to a project or activity;
(2) Individuals involved can be specifically identified with the project or activity;
(3) Such costs are explicitly included in the budget or have the prior written approval of the Federal awarding agency; and
(4) The costs are not also recovered as indirect costs.”
The meaning of (4) is the same as that of 200.403(d) above. That is to say, the project must require support services beyond the normal scope necessary for the typical sponsored project (i.e., it is an unlike circumstance).
Personnel may be hired to work on a sponsored project on an hourly basis for periods up to 12 months. Individuals hired on this basis receive no staff benefits other than Social Security and should be advised accordingly. The Personnel Service Center should be consulted to obtain the appropriate hourly rates for various categories of employment.
Staff benefits are charged to sponsored project accounts on a real cost basis. Depending on the mix of personnel assigned to the project, the staff benefit rate may show significant variation. While it may be possible to apply an average benefit rate (30%), it may be more appropriate in some situations to calculate the staff benefits on an employee-by-employee basis. The range of applicable benefit rates are provided in Staff Benefits Table .
GSRA Compensation, Tuition, and Benefits
GSRA Cost Estimates are for budget estimating purposes only and may vary from school to school. A GSRA appointment may be held from May through August, even though the GSRA is not enrolled in the University during that time. If the appointment is for the winter and fall terms, the fringe benefit charges should be budgeted for the full year, since the student is eligible for coverage during the intervening summer even though he or she is not on a GSRA appointment at that time.
The non-resident tuition differential is provided by the University for out-of-state students appointed as GSRAs. In-state tuition should be charged to sponsored accounts for GSRAs with appointments of 25% or greater. However, Schools and Colleges may provide tuition fellowships to cover a portion of the in-state tuition for GSRAs (see GSRA Cost Estimates ). The portion of the in-state tuition that remains after the fellowship is applied must be included on the grant as a charge to the sponsor.
In-state tuition charges should not be included as part of the GSRA stipend. The modified total direct cost (MTDC) base on which the University's indirect cost rate is calculated must exclude tuition charges. Therefore, indirect costs are not recoverable on tuition charges included in proposals for which the indirect cost rate is based on MTDC. Please contact the appropriate ORSP Project Representative should you have any questions.
Consumable Supplies and Materials
Consumable supplies are items used exclusively in support of project objectives. If it can be demonstrated that such supplies are used only in the conduct of the project and not for other purposes and are consumed completely in the course of the project, such items can be included as direct costs. Laboratory supplies, laboratory notebooks, printer paper for research data and reports, and so forth usually can be justified as consumable supplies. However, when supply items are purchased to support the multiple activities of project personnel, they are considered office supplies and cannot be charged directly to federal funds. Such items would include University stationary, pens, tablets, file folders, staples, paper clips, etc.
The estimated costs of consumable supplies and materials should be indicated in the proposed budget. It is generally acceptable to sponsors to provide a breakdown of supplies and materials by broad categories as opposed to the detailed listing of individual items. Contracts awarded by industries holding a prime contract with a federal agency, however, may require detailed itemization of supplies.
Major items of equipment proposed for acquisition should be itemized by descriptive name and estimated cost, and an adequate justification should be provided in the proposal narrative. Items costing less than $5000 or with a useful life expectancy of less than one year normally should be included under "Supplies and Materials." Shipping and/or installation charges associated with equipment acquisitions should be included in the cost estimates but generally are not itemized.
Specialized Services
Charges for computing services should be budgeted whenever these costs are justified. It is essential, however, that the budget clearly differentiate between central computing services provided by Information Technology Services and other computing services.
Other specialized service centers that have an approved user rate should be included in the proposed budget on a cost basis that reflects the recharge rates with the anticipated number of hours or other units of service clearly indicated. Once established, the schedule of rates must be applied to all users of the services/facilities, including internal-university users. Recharge rates are designed to recover, over the long term, not more than the aggregate cost of the services provided. The recharge/user rate should be included as part of the modified total direct cost (MTDC) for the project and should carry the appropriate indirect cost rate .
Consultants and Subcontracts
Federal agencies frequently establish a maximum daily rate of pay for consultants--specific dollar limits for various agencies are available from ORSP. The University must enter into a formal agreement with the consultant prior to the initiation of his or her effort. Consultant agreements as subject to the full recovery of indirect costs at the rate applicable to other direct cost items in the proposed budget.
The entire cost of a subcontract is normally shown as a single line item under "Other Direct Costs." A formal proposal from the subcontractor--including a statement of work, a detailed budget, period of performance, and key personnel--should be included to support this cost element. The Project Director should provide an explanation of why and how the proposed sub-contractor was selected, including the number of bids obtained.
Subcontracted effort requires a formal agreement between the University and the subcontractor, signed by a University official authorized to enter into contractual agreements on behalf of the Board of Regents. See: the Subcontracts and Hybrid Purchase Orders web page for criteria and procedures. Indirect costs are recovered on the first $25,000 of each subcontract.
Indirect Costs
Indirect costs are real costs of University operations that are not readily assignable to a particular project. The sample budget illustrates the procedures for applying the indirect cost rate .
Human Subject Fees
Human subjects often are paid a fee for their participation in research projects. Protocols for the use of human beings in research, teaching, or testing are reviewed and approved, according to federal, state and university policies. (See: Human Research Protection Program )
Other Costs
Funds may be requested from the sponsor to cover travel costs associated with the proposed project. Sponsors often require a breakdown of such travel costs by trip, reflecting the purpose, point of travel, number of persons, number of days, air fare, lodging and meal costs (per diem), and so forth. If foreign travel is contemplated, the proposal should include relevant information (including names of countries to be visited) and justification. Some sponsors have special regulations (e.g., use of domestic air carriers) governing foreign travel.
Costs of preparing and publishing reports of project results should be included in proposed budgets. Since page charges often are billed well after the completion of the research, it may be necessary to secure time extensions to pay these charges prior to the time that the project is closed out.
Other anticipated direct costs should be itemized--for example, equipment rental, maintenance agreements, or off-campus space rental. Telephone services and postage should not be included unless these costs are expected to be major elements in the project (e.g., telephone surveys). "Miscellaneous" or "contingency" categories should not be included. Items normally considered indirect costs should not be included in the proposed budget unless they are extraordinary and for unlike circumstances (e.g., utility costs required to operate a high-energy particle accelerator).
Network costs, including the hardware, software, personnel services, public access sites, and other related costs required to enable University personnel to share software or data or to communicate electronically with other individuals, are generally considered to be part of the physical infrastructure of the University and should not be included as direct costs in the proposal budget, as these costs are indirect in nature and included as a component of the Facilities & Administrative rate. However, individual workstations and specialized hardware and software attached to the network, which are not available to all users, are not included as part of the network costs and therefore may be treated as direct costs and recovered from sponsored projects through the use of approved recharge rates.
Questions regarding the appropriate treatment of network costs as either direct costs or Facilities & Administrative costs in proposal budgets should be forwarded to the Office of Research and Sponsored Projects.
References and Resources
- Direct Cost Other than Salaries
- Graduate Student Salaries (GSRAs)
- Staff Fringe Benefits
- Rackham Cost Sharing for Federal Training Grants
- Sample Budget Table
- Contact your unit-level research administrator for the best practices for your department, school, college, or unit.
- Contact your ORSP Project Representative regarding any questions about University policies and procedures.
- Contact the Office of Contract Administration for further information on subcontracts and consulting agreements.
- For information on how to pay human subjects, please see the Treasurer's Office requester/study-coordinator resources and the HSIP (Human Subject Incentive Program .
- Budget Reallocation Process (formerly 7471)
- Direct Costs
- Facilities and Administrative Costs (F&A)
- HSIP (Human Subjects Incentives Program)
Sample Budget Justifications
Sponsor requirements differ, and sample budget justifications should be seen only as a starting point. Guidelines for sponsor requirements are in the annotated budget justifications. Read the solicitation and the sponsor’s proposal preparation guidelines for each proposal's requirements.
For Research Sponsors
- Sample Budget Justification for Non-Federal Research [DOCX] - April 6, 2023
- Annotated Budget Justification - Non-Federal Research
- Sample Budget Justification for Federal Research [DOCX] - April 6, 2023
- Annotated Budget Justification - Federal Research
For Non-Research Sponsors:
- Sample Budget Justification for Non-Federal Non-Research [DOCX] - July 29, 2022
- Annotated Budget Justification - Non-Federal Non-Research
- Sample Budget Justification for Federal Non-Research [DOCX] - July 29, 2022
- Annotated Budget Justification - Federal Non-Research
- Uniform Guidance Fixed Rate Requirements
- F&A Methodology
- F&A Components
- MIT Use of a de minimis Rate
- Fund Account Overhead Rates
- Allocation Rates
- Determination of On-Campus and Off-Campus Rates
- Employee Benefits (EB) Rates
- Vacation Accrual Rates
- Graduate Research Assistant Tuition Subsidy
- Historical RA Salary Levels
- MIT Facts and Profile Information
- Classification of Sponsored Projects
- Types of Sponsored Awards
- How Are Sponsored Projects Generated?
- Cost Principles and Unallowable Costs
- Direct and Indirect Costs
- Pre-Proposals / Letters of Intent
- MIT Investigator Status
- Components of a Proposal
- Special Reviews
- Applying Through Workspace
- Proposal Preparation Checklist
- Proposals and Confidential Information
- Personnel Costs
- Subcontracts and Consultants
- Kuali Coeus Approval Mapping
- Roles and Responsibilities
- Submission of Revised Budgets
- Standard Contract Terms and Conditions
- Contractual Obligations and Problematic Terms and Conditions
- Review and Negotiation of Federal Contract and Grant Terms and Conditions
- Industrial Collaboration
- International Activities
- MIT Export Control - Export Policies
- Nondisclosure and Confidentiality Agreements
- Negative Confirmation On Award Notices
- Routing and Acceptance of the Award Notice
- COI and Special Review Hold Notice Definitions
- Limiting Long-Term WBS Account Structures
- SAP Project WBS Element Conditions
- Kuali Coeus Electronic Document Storage (EDS)
- Billing Agreements
- PI Absence from Project
- Cost Transfers
- Equipment Threshold
- Uniform Guidance and the FAR
- MIT Standard Terms and Policies
- Guidelines for Charging Faculty Summer Salary
- Key Personnel
- Limitations on Funds - Federal Contracts
- Managing Salary Costs
- Monitoring Project Budgets
- Monthly Reconciliation and Review
- No-Cost Extensions
- Reporting Requirements
- Return of Unexpended Funds to Foundations
- Determining the Sponsor Approved Budget (SAB)
- Working With the Sponsor Approved Budget (SAB)
- Sponsor Approved Budget (SAB) and Child Account Budgets
- Sponsor Approved Budget (SAB) and Prior Approvals
- Submitting an SAB Change Request
- When a PI Leaves MIT
- Research Performance Progress Reports
- Closing Out Fixed Price Awards
- Closeout of Subawards
- Record Retention
- Early Termination
- Reporting FAQs
- Using SciENcv
- AFOSR No-Cost Extension Process
- Terms and Conditions
- New ONR Account Set Ups
- Department of Defense Disclosure Guidance
- Department of Energy / Office of Science Disclosure Guidance
- Introduction to Industrial Sponsors
- General Considerations for Industrial Proposals
- SRC Guidance to Faculty Considering Applying for SRC Funding
- Find Specific RFP Information
- Industrial Proposal Checklist
- Proposal Formats
- Special Requirements
- Deadline Cycles
- Model Proposals
- Non-Competitive Industrial Proposals
- Master and Alliance Agreements With Non-Standard Proposal Processes
- Template Agreements
- New Consortium Agreements
- Competitive Industrial Proposals
- Collaborative (No-cost) Research Agreements
- National Aeronautics and Space Administration Disclosure Guidance
- NASA Graduate Research Fellowship Programs
- NASA PI Status and Definitions
- NIH Checklists and Preparation Guides
- National Institutes of Health Disclosure Guidance
- Human Subjects and NIH Proposals
- NIH Data Management and Sharing
- NIH Research Performance Progress Reports
- Grant Opportunities for Academic Liaison with Industry (GOALI) proposals
- MIT Guidance Regarding the NSF CAREER Program
- Research Experiences for Undergraduates (REU) Supplements
- National Science Foundation Disclosure Guidance
- NSF Proposals: Administrative Review Stage
- NSF Collaborations
- NSF Pre-Award and Post-Award Actions
- NSF Reporting
- NSF Frequently Asked Questions
- NSF Safe and Inclusive Working Environment
- Process, Roles and Responsibilities
- What Is Allowable/Eligible Cost Sharing?
- MIT’s Preferred Cost Sharing Funds
- Third-Party Cost Sharing
- Showing Cost Sharing in a Proposal Budget
- Sponsor Specific Instructions Regarding Location in the Proposal
- Funding F&A Costs as Cost Sharing
- Using Faculty Effort for Cost Sharing
- Information about Completing the Cost Sharing Template
- NSF Cost Sharing Policy
- Tracking/Reporting Cost Sharing
- Special Cost Sharing Topics
- International Activities Examples
- Rubicon Fellowships
- Marie Skłodowska-Curie Fellowships
- Criteria for Subrecipients
- Subawards at Proposal
- Requesting New Subawards
- Managing Subawards
- RAS Subaward Team Contacts
- Funding and Approval
- Proposal Phase
- Award Set-up
- Monitoring Research During Project Period
- Closeout Phase
- Voluntary Cost Sharing
- Sponsor-Specific Guidance
- Audits and Auditors
- Upcoming Trainings and Events
- Research Administration Practices (RAP)
- NCURA Virtual Workshops and Webinars
- Guide to RA Resources and Training
- Career Paths
- Newsletters
- Tools and Systems
- Award Closeout & Audits
- Award Setup
- Cost Sharing
- Export Control
- Financial Conflict of Interest
- Kuali Coeus
- Project Monitoring
- Proposal Preparation & Submission
- Research Sub Awards
- Research Administration Email Lists
- RAS Operations
- VPR Research Administration Organization Chart
- By department
- By administrator
- Research Administrator Day
- News & Announcements
- Onsite searching on the VPR public websites
| --> |
| > > to return to Guidelines page |
| | © RWJF 2008 | | |

Qualitative Research Proposal
Proposal maker.

Writing a qualitative research proposal is just like writing any other research proposals. The only thing is that you are writing specifically designed to provide non-numerical data, concepts and the like. You are more likely to follow a specific format since it is a type of academic writing.
6+ Qualitative Research Proposal Examples
1. qualitative research proposal gantt chart template.
2. Sample Qualitative Research Proposal

Size: 90 KB
3. Proposal in Qualitative Research Template

Size: 15 KB
4. Individual Qualitative Research Proposal

5. Qualitative Research Proposal Format

Size: 517 KB
6. Elements of Research Proposal Qualitative Design

Size: 23 KB
7. Qualitative Research Workshop Proposal

Size: 559 KB
What is a Qualitative Research Proposal?
A qualitative research proposal gives the detailed summary of your research study. It is a type of research proposal that only involves qualitative methods of gathering a certain data such as an interview, observation, questionnaire, or case studies . Qualitative research can be applied in the field of psychology, social sciences and the like.
How to Write a Qualitative Research Proposal?
Think of a unique topic for you to provide a good research title.
Example: A Qualitative Study on Coping up with the Different Levels of Anxiety among Students
Develop Research Questions
Your research questions will be your guide in your research study. It contains the research design, research methodology and the technique you used in collecting data.
Example: What do the architecture and engineering students with anxiety do to cope up with their studies in the university?
For qualitative research, we can use the SPIDER method which stands for Sample, Phenomenon of Interest, Design, Evaluation and Research type.
Sample refers to your target population that is included in your study.
Example: The population consisted of a community of architecture and engineering students of the oldest university in the city of Manila, Philippines.
Phenomenon of Interest refers to an event or an object. What could be their experience in the university?
Design refers to the methods you used in conducting the study.
- Interview – refers to the one on one interaction with the participant.
- Observation – refers to observing the participants whether or not they are fully aware of the thought that you are observing them.
- Questionnaire – refers to the process of distributing survey questionnaires to gather answers from your participants. It ends with tallying the answers to see what the participants choose the most.
- Case study – refers to an intensive study about a specific person or group of people.
Ensure That Some Ethical Standards are Met
This refers to protecting the privacy or confidentiality of the data you have gathered and the rights of the participants.
“There were more ethical considerations in almost all aspects for drug trials and clinical studies compared with proposals for epidemiological studies. Clinical research studies usually directly involve human subjects, either with preventive, therapeutic, or non-therapeutic procedures. In general, the study procedures in such study designs put human subjects at higher risks, thus there are more ethical concerns. The primary ethical considerations of clinical studies are competent medical treatment and care, alongside an acceptable risk–benefit balance. However, many laboratory research studies use stored specimens, with less invasive procedures, and epidemiology studies usually employ data collection through medical records, CRFs or questionnaires. Ethical issues for the latter, therefore, mainly concern confidentiality and privacy of the study participants. However, it was found that studies that collect new specimens received more comments on ethical issues. There remains debate among RECs about solutions for issues around sample export, storage, and reuse. However, it is recommended that in order to ensure adequate protection of human research subjects participating in scientific research, RECs bear the responsibility of guaranteeing that participants are provided with sufficient detail to be able to provide informed consent as well as to understand the reality of genetic research as it is practiced.”
www.ncbi.nlm.nih.gov
Consider writing a plan to be used for the whole duration of your research. this includes the timeline and budget..
Timeline – refers to the target length of time to complete your research.
Budget – refers to the estimation of how much your research would cost. All items that you think might be included in the budgeting must be included.
Don’t Forget to Include Your Reference
This contains the list of the sources that you should cite on the last page of your research. It usually follows the APA format.
How long should a qualitative research proposal be?
Every research proposal should be at least 4 to 7 pages long or depending on the requirement of your professor.
Do we still have to write for the definition of terms in the research proposal?
Yes. You have the option to do so to introduce and define words that are difficult for the readers to understand.
What can be considered as a good topic in writing qualitative research?
Your topic will either be given by your professor or you may look into unique topics into the internet.
Text prompt
- Instructive
- Professional
Generate a proposal for a new school recycling program
Compose a proposal for a school field trip to a science museum.
- Environment
- Science & Technology
- Business & Industry
- Health & Public Welfare
- Topics (CFR Indexing Terms)
- Public Inspection
- Presidential Documents
- Document Search
- Advanced Document Search
- Public Inspection Search
- Reader Aids Home
- Office of the Federal Register Announcements
- Using FederalRegister.Gov
- Understanding the Federal Register
- Recent Site Updates
- Federal Register & CFR Statistics
- Videos & Tutorials
- Developer Resources
- Government Policy and OFR Procedures
- Congressional Review
- My Clipboard
- My Comments
- My Subscriptions
- Sign In / Sign Up
- Site Feedback
- Search the Federal Register
The Federal Register
The daily journal of the united states government.
- Legal Status
This site displays a prototype of a “Web 2.0” version of the daily Federal Register. It is not an official legal edition of the Federal Register, and does not replace the official print version or the official electronic version on GPO’s govinfo.gov.
The documents posted on this site are XML renditions of published Federal Register documents. Each document posted on the site includes a link to the corresponding official PDF file on govinfo.gov. This prototype edition of the daily Federal Register on FederalRegister.gov will remain an unofficial informational resource until the Administrative Committee of the Federal Register (ACFR) issues a regulation granting it official legal status. For complete information about, and access to, our official publications and services, go to About the Federal Register on NARA's archives.gov.
The OFR/GPO partnership is committed to presenting accurate and reliable regulatory information on FederalRegister.gov with the objective of establishing the XML-based Federal Register as an ACFR-sanctioned publication in the future. While every effort has been made to ensure that the material on FederalRegister.gov is accurately displayed, consistent with the official SGML-based PDF version on govinfo.gov, those relying on it for legal research should verify their results against an official edition of the Federal Register. Until the ACFR grants it official status, the XML rendition of the daily Federal Register on FederalRegister.gov does not provide legal notice to the public or judicial notice to the courts.
Agency Information Collection Activities; Submission for Office of Management and Budget Review; Comment Request; Generic Clearance for the Collection of Qualitative Data on Tobacco Products and Communications
A Notice by the Food and Drug Administration on 06/18/2024
This document has a comment period that ends in 27 days. (07/18/2024) Submit a formal comment
Thank you for taking the time to create a comment. Your input is important.
Once you have filled in the required fields below you can preview and/or submit your comment to the Health and Human Services Department for review. All comments are considered public and will be posted online once the Health and Human Services Department has reviewed them.
You can view alternative ways to comment or you may also comment via Regulations.gov at https://www.regulations.gov/commenton/FDA-2014-N-0987-0024 .
- What is your comment about?
Note: You can attach your comment as a file and/or attach supporting documents to your comment. Attachment Requirements .
this will NOT be posted on regulations.gov
- Opt to receive email confirmation of submission and tracking number?
- Tell us about yourself! I am... *
- First Name *
- Last Name *
- State Alabama Alaska American Samoa Arizona Arkansas California Colorado Connecticut Delaware District of Columbia Florida Georgia Guam Hawaii Idaho Illinois Indiana Iowa Kansas Kentucky Louisiana Maine Maryland Massachusetts Michigan Minnesota Mississippi Missouri Montana Nebraska Nevada New Hampshire New Jersey New Mexico New York North Carolina North Dakota Ohio Oklahoma Oregon Pennsylvania Puerto Rico Rhode Island South Carolina South Dakota Tennessee Texas Utah Vermont Virgin Islands Virginia Washington West Virginia Wisconsin Wyoming
- Country Afghanistan Åland Islands Albania Algeria American Samoa Andorra Angola Anguilla Antarctica Antigua and Barbuda Argentina Armenia Aruba Australia Austria Azerbaijan Bahamas Bahrain Bangladesh Barbados Belarus Belgium Belize Benin Bermuda Bhutan Bolivia, Plurinational State of Bonaire, Sint Eustatius and Saba Bosnia and Herzegovina Botswana Bouvet Island Brazil British Indian Ocean Territory Brunei Darussalam Bulgaria Burkina Faso Burundi Cambodia Cameroon Canada Cape Verde Cayman Islands Central African Republic Chad Chile China Christmas Island Cocos (Keeling) Islands Colombia Comoros Congo Congo, the Democratic Republic of the Cook Islands Costa Rica Côte d'Ivoire Croatia Cuba Curaçao Cyprus Czech Republic Denmark Djibouti Dominica Dominican Republic Ecuador Egypt El Salvador Equatorial Guinea Eritrea Estonia Ethiopia Falkland Islands (Malvinas) Faroe Islands Fiji Finland France French Guiana French Polynesia French Southern Territories Gabon Gambia Georgia Germany Ghana Gibraltar Greece Greenland Grenada Guadeloupe Guam Guatemala Guernsey Guinea Guinea-Bissau Guyana Haiti Heard Island and McDonald Islands Holy See (Vatican City State) Honduras Hong Kong Hungary Iceland India Indonesia Iran, Islamic Republic of Iraq Ireland Isle of Man Israel Italy Jamaica Japan Jersey Jordan Kazakhstan Kenya Kiribati Korea, Democratic People's Republic of Korea, Republic of Kuwait Kyrgyzstan Lao People's Democratic Republic Latvia Lebanon Lesotho Liberia Libya Liechtenstein Lithuania Luxembourg Macao Macedonia, the Former Yugoslav Republic of Madagascar Malawi Malaysia Maldives Mali Malta Marshall Islands Martinique Mauritania Mauritius Mayotte Mexico Micronesia, Federated States of Moldova, Republic of Monaco Mongolia Montenegro Montserrat Morocco Mozambique Myanmar Namibia Nauru Nepal Netherlands New Caledonia New Zealand Nicaragua Niger Nigeria Niue Norfolk Island Northern Mariana Islands Norway Oman Pakistan Palau Palestine, State of Panama Papua New Guinea Paraguay Peru Philippines Pitcairn Poland Portugal Puerto Rico Qatar Réunion Romania Russian Federation Rwanda Saint Barthélemy Saint Helena, Ascension and Tristan da Cunha Saint Kitts and Nevis Saint Lucia Saint Martin (French part) Saint Pierre and Miquelon Saint Vincent and the Grenadines Samoa San Marino Sao Tome and Principe Saudi Arabia Senegal Serbia Seychelles Sierra Leone Singapore Sint Maarten (Dutch part) Slovakia Slovenia Solomon Islands Somalia South Africa South Georgia and the South Sandwich Islands South Sudan Spain Sri Lanka Sudan Suriname Svalbard and Jan Mayen Swaziland Sweden Switzerland Syrian Arab Republic Taiwan, Province of China Tajikistan Tanzania, United Republic of Thailand Timor-Leste Togo Tokelau Tonga Trinidad and Tobago Tunisia Turkey Turkmenistan Turks and Caicos Islands Tuvalu Uganda Ukraine United Arab Emirates United Kingdom United States United States Minor Outlying Islands Uruguay Uzbekistan Vanuatu Venezuela, Bolivarian Republic of Viet Nam Virgin Islands, British Virgin Islands, U.S. Wallis and Futuna Western Sahara Yemen Zambia Zimbabwe
- Organization Type * Company Organization Federal State Local Tribal Regional Foreign U.S. House of Representatives U.S. Senate
- Organization Name *
- You are filing a document into an official docket. Any personal information included in your comment text and/or uploaded attachment(s) may be publicly viewable on the web.
- I read and understand the statement above.
- Preview Comment
Document Details
Information about this document as published in the Federal Register .
Document Statistics
Enhanced content.
Relevant information about this document from Regulations.gov provides additional context. This information is not part of the official Federal Register document.
Published Document
This document has been published in the Federal Register . Use the PDF linked in the document sidebar for the official electronic format.
Enhanced Content - Table of Contents
This table of contents is a navigational tool, processed from the headings within the legal text of Federal Register documents. This repetition of headings to form internal navigation links has no substantive legal effect.
FOR FURTHER INFORMATION CONTACT:
Supplementary information:, generic clearance for the collection of qualitative data on tobacco products and communications, omb control number 0910-0796—extension, enhanced content - submit public comment.
- Submit a public comment on this document
Enhanced Content - Read Public Comments
- This feature is not available for this document.
Enhanced Content - Sharing
- Email this document to a friend
Enhanced Content - Document Print View
- Print this document
Enhanced Content - Document Tools
These tools are designed to help you understand the official document better and aid in comparing the online edition to the print edition.
These markup elements allow the user to see how the document follows the Document Drafting Handbook that agencies use to create their documents. These can be useful for better understanding how a document is structured but are not part of the published document itself.
Enhanced Content - Developer Tools
This document is available in the following developer friendly formats:.
- JSON: Normalized attributes and metadata
- XML: Original full text XML
- MODS: Government Publishing Office metadata
More information and documentation can be found in our developer tools pages .
Official Content
- View printed version (PDF)
This PDF is the current document as it appeared on Public Inspection on 06/17/2024 at 8:45 am. It was viewed 0 times while on Public Inspection.
If you are using public inspection listings for legal research, you should verify the contents of the documents against a final, official edition of the Federal Register. Only official editions of the Federal Register provide legal notice of publication to the public and judicial notice to the courts under 44 U.S.C. 1503 & 1507 . Learn more here .
Food and Drug Administration, HHS.
The Food and Drug Administration (FDA, us, or we) is announcing that a proposed collection of information has been submitted to the Office of Management and Budget (OMB) for review and clearance under the Paperwork Reduction Act of 1995.
Submit written comments (including recommendations) on the collection of information by July 18, 2024.
To ensure that comments on the information collection are received, OMB recommends that written comments be submitted to https://www.reginfo.gov/public/do/PRAMain . Find this particular information collection by selecting “Currently under Review—Open for Public Comments” or by using the search function. The OMB control number for this information collection is 0910-0796. Also include the FDA docket number found in brackets in the heading of this document.
JonnaLynn Capezzuto, Office of Operations, Food and Drug Administration, Three White Flint North, 10A-12M, 11601 Landsdown St., North Bethesda, MD 20852, 301-796-3794, [email protected] .
In compliance with 44 U.S.C. 3507 , FDA has submitted the following proposed collection of information to OMB for review and clearance.
This information collection supports FDA's programs. Under section 1003(d)(2)(D) of the Federal Food, Drug, and Cosmetic Act ( 21 U.S.C. 393(d)(2)(D) ), FDA is authorized to conduct educational and public information programs.
In conducting studies relating to the regulation and communications related to tobacco products, FDA will need to employ formative qualitative research including but not limited to focus groups, usability and/or psychometric testing, in-depth interviews (IDIs), cognitive interviews and asynchronous qualitative discussions ( e.g., online journaling or web-based discussion boards), naturalistic observation and ethnographic studies to assess knowledge and perceptions about tobacco-related topics with specific target audiences. The information collected will serve four major purposes. First, foundational research will provide critical knowledge and insights about intended audiences. FDA must first understand people's knowledge of, perceptions of, and reactions to tobacco related topics prior to developing survey/research questions as well as stimuli for experimental studies. Second, formative research will provide information about people's responses, thoughts, and feelings regarding potential creative messaging, or stimuli. Third, by collecting communications usability information, FDA will be able to serve and respond to the ever-changing demands of consumers of tobacco products. Additionally, we will be able to determine the best way to communicate with intended audiences around tobacco prevention and cessation. Fourth, cognitive testing will allow FDA to assess consumer understanding of survey/research questions and study stimuli. Focus groups and/or IDIs with a sample of the intended audience will allow FDA to refine the survey/research questions and study stimuli while they are still in the developmental stage. FDA will collect, and interpret information gathered through this generic clearance to: (1) better understand characteristics of the intended audience—its perceptions, knowledge, attitudes, beliefs, and behaviors—and use these in the development of appropriate survey/research questions, study stimuli, or communications; (2) more efficiently and effectively design survey/research questions and study stimuli; and (3) more efficiently and effectively design experimental studies.
FDA is requesting approval of an extension of this generic clearance for collecting information using qualitative methods ( e.g., interviews, focus groups, asynchronous discussion boards, etc.) for studies involving all tobacco products regulated by FDA. This information will be used to explore concepts of interest and assist in the development of quantitative study proposals, complementing other important research efforts in the Agency. This information may also be used to help identify and develop communication messages, which may be used in education campaigns. Qualitative research plays an important role in gathering information because it allows for an in-depth understanding of Start Printed Page 51535 individuals' attitudes, beliefs, motivations, and feelings. Qualitative research serves the narrowly defined need for direct and informal public opinion on a specific topic.
The number of respondents to be included in each new study may vary, depending on the nature of the study ( e.g., foundational, formative, etc.), approach (synchronous vs. asynchronous, or virtual vs. in person) and the intended audience. Table 1 provides examples of the types of studies that may be administered and estimated burden levels during the 3-year period. Time to read, view, or listen to the message being tested is built into the “Average Burden per Response” figures.
In accordance with 5 CFR 1320.8(d) , FDA published a 60-day notice for public comment on the proposed collection of information in the Federal Register of January 9, 2024 ( 89 FR 1097 ). FDA received two PRA related comments.
( Comment ) The comment expressed that the Paperwork Reduction Act was written to reduce burden on the public, but the overuse of surveys is encouraged generically, making it even easier to collect information with no need based. The comment stated further that “This seems to be counterintuitive to the purpose of the PRA and exactly what the Act was supposed to be protecting us from, another survey we do not have time or resources to complete buy you would like to give us with no specific goal. Overuse.”
( Response ) FDA disagrees with the comment suggesting that the generic information collection process enables the overuse of surveys and undermines the Paperwork Reduction Act. In response to this comment, FDA has updated Supporting Statement Part A to clarify the necessity of the information collected under this generic clearance for the proper performance of FDA CTP's function and the practical utility of collecting such information. The information collected will support FDA CTP's function by advancing CTP's Strategic Plan and its specific goals: “Goal 1: Develop, Advance, and Communicate Comprehensive and Impactful Tobacco Regulations and Guidance” and “Goal 4: Enhance Knowledge and Understanding of the Risks Associated with Tobacco Product Use.” The practical utility of the collected data is evidenced by its role in facilitating the development of clear and accessible CTP public statements and communications, such as web content, press releases, fact sheets, and retailer resources. Furthermore, the utility is demonstrated by CTP achieving the following objectives with specific audiences:
- Educating youth about the risks of tobacco product use.
- Educating people who use tobacco products about the benefits of cessation.
- Educating adults who smoke about the relative risks of tobacco products.
This foundational research has helped FDA to understand audiences and inform message development and the testing of messages in communicating the risks of tobacco use, how to quit using tobacco products, and FDA's role in regulating tobacco. Obtaining this information has allowed FDA to improve messages, materials and implementation strategies while revisions are still affordable and possible.
(Comment) The comment expressed the lack of specificity regarding FDA's public education goals. Specifically, the comment notes that FDA vaguely states it will collect qualitative data to “explore concepts of interest and assist in the development of quantitative research proposals” and “help identify and develop communication messages, which may be used in education campaigns.” The comment stated further that they encourage “FDA to prioritize educating adults, particularly adult smokers and physicians and medical staff who advise adult smokers about tobacco harm reduction. [. . .], FDA's public education campaigns should aim to improve understanding among adult smokers where there currently exists significant uncertainty and confusion about materially important issues that are detrimental to public-health efforts. These important issues include “educating the adult public, particularly adult smokers, about the continuum of risk, and where alternatives to combustible cigarettes fall on that continuum,” and “correcting misunderstandings about the absence of any direct causal link between nicotine and tobacco-related diseases.”
(Response) FDA disagrees with the comment suggesting that an overarching purpose or plan for communications, information goals, or target audiences was not provided. In response to this comment, FDA has updated Supporting Statement Part A to clarify that the information collected under this generic clearance is necessary for the proper performance of FDA CTP's function and will be of practical utility in advancing CTP's Strategic Plan and its specific goals. The information collected under this generic clearance will advance CTP's objectives to educate people who use tobacco products about the benefits of cessation and to educate adults who smoke about the relative risks of tobacco products. The following generic information collections were recently approved under 0910-0796. FDA has summarized how they address specific objectives such as educating adults about tobacco products' relative risks:
- “Consumer Perceptions of Cessation and Harm”: Focus group study with established cigarette smokers ages 25 and up. The objective is to learn about consumer perceptions (and misconceptions) regarding nicotine and tobacco products.
- “Consumer Perceptions of Modified and Reduced Risk (MoRR)”: Focus group study with current and established cigarette smokers ages 21 and older. The objective is to gain information to inform health communication materials dedicated to modified risk products and/or the continuum of risk. This may help reduce misperceptions and lack of awareness.
- “Menthol User Audience Research”: In-depth interviews conducted with adult menthol smokers ages 21 and older. The objective is to examine demographic, sociocultural, psychographic, and behavioral characteristics of adult menthol cigarette users; to identify segments that are most likely to adopt less harmful behaviors in response to targeted messaging; and to identify communication strategies to support menthol smokers in adopting less harmful behaviors.
- “Qualitative Study of Product Category Comparison Statements—modified risk tobacco product (MRTPs) and harmful and potentially harmful constituents (HPHCs)”: Focus group study with adult current and former cigarette smokers ages 18 and older. The objective is to understand participants' knowledge, attitudes, beliefs, and perceptions about different tobacco products. This includes a stimulus-driven discussion of HPHC information and MRTP claims.
FDA estimates the burden of this collection of information as follows: Start Printed Page 51536
| Type of interview | Number of respondents | Number of responses per respondent | Total annual responses | Average burden per response | Total hours |
|---|---|---|---|---|---|
| In-Person Individual In-depth Interviews | 4,500 | 1 | 4,500 | 1 | 4,500 |
| In-depth Interview Screener | 22,500 | 1 | 22,500 | 0.083 (5 minutes) | 1,875 |
| Focus Group Screener | 56,000 | 1 | 56,000 | 0.25 (15 minutes) | 14,000 |
| Focus Group Discussion | 252,000 | 1 | 252,000 | 1.5 | 378,000 |
| Discussion Board Screener | 8,000 | 1 | 8,000 | 0.083 (5 minutes) | 667 |
| Discussion Board Participation | 100 | 1 | 100 | 1.5 | 150 |
| Total | 399,192 | ||||
| There are no capital costs or operating and maintenance costs associated with this collection of information. | |||||
Our estimated burden for the information collection reflects an overall increase of 384,258 hours and a corresponding increase of 314,926 responses. We attribute this adjustment to the number of study responses used during the current approval and now estimated for the next 3 years. A greater number of qualitative studies will be conducted over the next 3 years due to the need to develop new creative messages and content. Recent years have seen a dramatic change in media. With the shift to digital media, FDA must adapt to communicate effectively in a digital environment. As digital tobacco use prevention/interventions are still in their infancy, we must better understand the types of digital channels available. To impact public health outcomes, we need to understand how to reach our intended audience. New foundational studies are needed (including those on digital metrics, measurement, and implementation). As a result, we have adjusted our burden estimate and revised the number of respondents to the information collection.
Dated: June 13, 2024.
Lauren K. Roth,
Associate Commissioner for Policy.
[ FR Doc. 2024-13386 Filed 6-17-24; 8:45 am]
BILLING CODE 4164-01-P
- Executive Orders
Reader Aids
Information.
- About This Site
- Accessibility
- No Fear Act
- Continuity Information

IMAGES
VIDEO
COMMENTS
This is Part 2 of a four-part series on proposal writing for qualitative research. Please read Part 1 of this series, which covers crafting an overview as a starting point of conversation with the…
A good budget shows the assessors that you have thought about your research in detail and, if it is done well, it can serve as a great, convincing overview of the project. Here are five steps to create a simple budget for your research project. 1. List your activities. Make a list of everything that you plan to do in the project, and who is ...
Proposal Budget Overview A budget is a financial proposal that reflects the work proposed. It outlines the expected project costs in detail and should mirror the project description. A budget is presented as a categorical list of anticipated project costs representing the researcher's best estimate of the funds needed to support the proposed work.
What is a Proposal Budget and why is it needed? Before we get started talking about all the pieces of a budget, let's make sure that we're on the same page about what a budget actually is. A budget is a financial proposal that reflects the work proposed. It outlines the expected project costs in detail, and should mirror the project description.
Budget and timeline . Figure 12.1 provides further detail of the content of each of these sections that require attention when ... When writing your qualitative research proposal the one thing you don't want to do is unintentionally mask your intent with impenetrable terminology. Remember a number of different reviewers will read your proposal
This is Part 1 of a four-part on proposal writing for qualitative research. Read the introduction for context on why we started this series. Qualitative research is full of nuanced details ...
Md. Ismail Hossain, Nafiul Mehedi, and Iftakhar Ahmad. Abstract The chapter discusses designing a research proposal in qualitative research. The main objective is to outline the major components of a qualitative research proposal with example(s) so that the students and novice scholars easily get an understanding of a qualitative proposal.
Breakdown of the budget into item-wise and year-wise with cost calculation should be done. It should be ensured that costs are reasonable, allowable and related to the research proposal, so that the budget appears realistic. Travel expenses should be calculated as per the rules of the funding agency.
However, as the original tool focused on quantitative research proposals, there was a need to adapt this tool specifically for qualitative research proposals. Qualitative research methods are increasingly recognized for their importance in healthcare-related research, particularly in contextualizing social and cultural realities that impact ...
• Qualitative research is often undertaken when little is known about a topic. This means a qualitative research proposal cannot be as clear in the detail as a quantitative one. Qualitative research is often exploratory and develops iteratively. It may be hard to specify what your outcomes are likely to be, beforehand.
The chapter discusses designing a research proposal in qualitative research. The main objective is to outline the major components of a qualitative research proposal with example (s) so that the students and novice scholars easily get an understanding of a qualitative proposal. The chapter highlights the major components of a qualitative ...
Creating a Budget. In general, while your research proposal outlines the academic significance of your study, the budget and budget narrative show that you have an understanding of what it will cost for you to be able to perform this research. Your proposed budget should identify all the expenses that are necessary and reasonable for the ...
A search was performed with keywords such as research proposal, funding, qualitative and writing proposals using search engines, namely, PubMed, Google Scholar and Scopus. Table 1. Five 'C's while writing a literature review. ... When the researcher prepares a research budget, he/she should predict and cost all aspects of the research and ...
Be the first to add your personal experience. 3. Choose your methods and tools wisely. Be the first to add your personal experience. 4. Estimate your costs realistically. Be the first to add your ...
Research proposal examples. Writing a research proposal can be quite challenging, but a good starting point could be to look at some examples. We've included a few for you below. Example research proposal #1: "A Conceptual Framework for Scheduling Constraint Management".
A research budget contains both direct costs and indirect costs (overhead), but the level of detail varies from sponsor to sponsor. The first step in developing a budget is to carefully read the guidelines of the funding opportunity being pursued. ... such as goods and/or services in support of a research project or proposal. It is challenging ...
The budgeting of your salary, a direct cost of the research, should be listed as $21,161.73. If your project covers three years, with the same or differing time commitments, you calculate this figure for each year of your project. Remember to factor in pay rises according to Step increases in multi-year grants.
A "starting date" should be specified, since it is essential to ensure accurate budget calculations. If cost-sharing is included, each budget period should include columns for both "Sponsor" and "University" costs. A budget summary should be included for proposals with multi-year funding. All budget entries should be rounded to the nearest ...
Sample Budget Justification NOTE: Prior to preparing a budget, the Principal Investigator, Research Grants Specialist and/or Office of ... The proposed project will utilize qualitative, as well as quantitative data requiring specialized analytical software. Funding for $1,250 is requested for <insert name of license; provide web or printed ...
Sample Budget Justifications. Sponsor requirements differ, and sample budget justifications should be seen only as a starting point. Guidelines for sponsor requirements are in the annotated budget justifications. Read the solicitation and the sponsor's proposal preparation guidelines for each proposal's requirements.
4. Calculate Income. After specifying your target audience, calculate your research's overall income. Include in your calculations the allotment of your research's financial assistance. Also, you have to indicate where these funds come from. The total income will be the very foundation of your budget. 5.
Budget. Qualitative research - data collection and analysis - is a time consuming enterprise that involves input from a knowledgable, multidisciplinary research team. Grant proposals should secure enough time and money for adequate training for those who will be collecting data. Training can be a time intensive process, if done properly.
What is a Qualitative Research Proposal? A qualitative research proposal gives the detailed summary of your research study. It is a type of research proposal that only involves qualitative methods of gathering a certain data such as an interview, observation, questionnaire, or case studies.Qualitative research can be applied in the field of psychology, social sciences and the like.
Qualitative research plays an important role in gathering information because it allows for an in-depth understanding of Start Printed Page 51535 individuals' attitudes, beliefs, motivations, and feelings. Qualitative research serves the narrowly defined need for direct and informal public opinion on a specific topic.