- The Open University
- Guest user / Sign out
- Study with The Open University

My OpenLearn Profile
Personalise your OpenLearn profile, save your favourite content and get recognition for your learning

Addressing ethical issues in your research proposal
This article explores the ethical issues that may arise in your proposed study during your doctoral research degree.
What ethical principles apply when planning and conducting research?
Research ethics are the moral principles that govern how researchers conduct their studies (Wellcome Trust, 2014). As there are elements of uncertainty and risk involved in any study, every researcher has to consider how they can uphold these ethical principles and conduct the research in a way that protects the interests and welfare of participants and other stakeholders (such as organisations).
You will need to consider the ethical issues that might arise in your proposed study. Consideration of the fundamental ethical principles that underpin all research will help you to identify the key issues and how these could be addressed. As you are probably a practitioner who wants to undertake research within your workplace, consider how your role as an ‘insider’ influences how you will conduct your study. Think about the ethical issues that might arise when you become an insider researcher (for example, relating to trust, confidentiality and anonymity).
What key ethical principles do you think will be important when planning or conducting your research, particularly as an insider? Principles that come to mind might include autonomy, respect, dignity, privacy, informed consent and confidentiality. You may also have identified principles such as competence, integrity, wellbeing, justice and non-discrimination.
Key ethical issues that you will address as an insider researcher include:
- Gaining trust
- Avoiding coercion when recruiting colleagues or other participants (such as students or service users)
- Practical challenges relating to ensuring the confidentiality and anonymity of organisations and staff or other participants.
(Heslop et al, 2018)
A fuller discussion of ethical principles is available from the British Psychological Society’s Code of Human Research Ethics (BPS, 2021).
You can also refer to guidance from the British Educational Research Association and the British Association for Applied Linguistics .

Ethical principles are essential for protecting the interests of research participants, including maximising the benefits and minimising any risks associated with taking part in a study. These principles describe ethical conduct which reflects the integrity of the researcher, promotes the wellbeing of participants and ensures high-quality research is conducted (Health Research Authority, 2022).
Research ethics is therefore not simply about gaining ethical approval for your study to be conducted. Research ethics relates to your moral conduct as a doctoral researcher and will apply throughout your study from design to dissemination (British Psychological Society, 2021). When you apply to undertake a doctorate, you will need to clearly indicate in your proposal that you understand these ethical principles and are committed to upholding them.
Where can I find ethical guidance and resources?
Professional bodies, learned societies, health and social care authorities, academic publications, Research Ethics Committees and research organisations provide a range of ethical guidance and resources. International codes such as the Universal Declaration of Human Rights underpin ethical frameworks (United Nations, 1948).
You may be aware of key legislation in your own country or the country where you plan to undertake the research, including laws relating to consent, data protection and decision-making capacity, for example, the Data Protection Act, 2018 (UK). If you want to find out more about becoming an ethical researcher, check out this Open University short course: Becoming an ethical researcher: Introduction and guidance: What is a badged course? - OpenLearn - Open University
You should be able to justify the research decisions you make. Utilising these resources will guide your ethical judgements when writing your proposal and ultimately when designing and conducting your research study. The Ethical Guidelines for Educational Research (British Educational Research Association, 2018) identifies the key responsibilities you will have when you conduct your research, including the range of stakeholders that you will have responsibilities to, as follows:
- to your participants (e.g. to appropriately inform them, facilitate their participation and support them)
- clients, stakeholders and sponsors
- the community of educational or health and social care researchers
- for publication and dissemination
- your wellbeing and development
The National Institute for Health and Care Research (no date) has emphasised the need to promote equality, diversity and inclusion when undertaking research, particularly to address long-standing social and health inequalities. Research should be informed by the diversity of people’s experiences and insights, so that it will lead to the development of practice that addresses genuine need. A commitment to equality, diversity and inclusion aims to eradicate prejudice and discrimination on the basis of an individual or group of individuals' protected characteristics such as sex (gender), disability, race, sexual orientation, in line with the Equality Act 2010.
The NIHR has produced guidance for enhancing the inclusion of ‘under-served groups’ when designing a research study (2020). Although the guidance refers to clinical research it is relevant to research more broadly.
You should consider how you will promote equality and diversity in your planned study, including through aspects such as your research topic or question, the methodology you will use, the participants you plan to recruit and how you will analyse and interpret your data.
What ethical issues do I need to consider when writing my research proposal?

You might be planning to undertake research in a health, social care, educational or other setting, including observations and interviews. The following prompts should help you to identify key ethical issues that you need to bear in mind when undertaking research in such settings.
1. Imagine you are a potential participant. Think about the questions and concerns that you might have:
- How would you feel if a researcher sat in your space and took notes, completed a checklist, or made an audio or film recording?
- What harm might a researcher cause by observing or interviewing you and others?
- What would you want to know about the researcher and ask them about the study before giving consent?
- When imagining you are the participant, how could the researcher make you feel more comfortable to be observed or interviewed?
2. Having considered the perspective of your potential participant, how would you take account of concerns such as privacy, consent, wellbeing and power in your research proposal?
[Adapted from OpenLearn course: Becoming an ethical researcher, Week 2 Activity 3: Becoming an ethical researcher - OpenLearn - Open University ]
The ethical issues to be considered will vary depending on your organisational context/role, the types of participants you plan to recruit (for example, children, adults with mental health problems), the research methods you will use, and the types of data you will collect. You will need to decide how to recruit your participants so you do not inappropriately exclude anyone. Consider what methods may be necessary to facilitate their voice and how you can obtain their consent to taking part or ensure that consent is obtained from someone else as necessary, for example, a parent in the case of a child.
You should also think about how to avoid imposing an unnecessary burden or costs on your participants. For example, by minimising the length of time they will have to commit to the study and by providing travel or other expenses. Identify the measures that you will take to store your participants’ data safely and maintain their confidentiality and anonymity when you report your findings. You could do this by storing interview and video recordings in a secure server and anonymising their names and those of their organisations using pseudonyms.
Professional codes such as the Code of Human Research Ethics (BPS, 2021) provide guidance on undertaking research with children. Being an ‘insider’ researching within your own organisation has advantages. However, you should also consider how this might impact on your research, such as power dynamics, consent, potential bias and any conflict of interest between your professional and researcher roles (Sapiro and Matthews, 2020).
How have other researchers addressed any ethical challenges?
The literature provides researchers’ accounts explaining how they addressed ethical challenges when undertaking studies. For example, Turcotte-Tremblay and McSween-Cadieux (2018) discuss strategies for protecting participants’ confidentiality when disseminating findings locally, such as undertaking fieldwork in multiple sites and providing findings in a generalised form. In addition, professional guidance includes case studies illustrating how ethical issues can be addressed, including when researching online forums (British Sociological Association, no date).
Watch the videos below and consider what insights the postgraduate researcher and supervisor provide regarding issues such as being an ‘insider researcher’, power relations, avoiding intrusion, maintaining participant anonymity and complying with research ethics and professional standards. How might their experiences inform the design and conduct of your own study?
Postgraduate researcher and supervisor talk about ethical considerations
Your thoughtful consideration of the ethical issues that might arise and how you would address these should enable you to propose an ethically informed study and conduct it in a responsible, fair and sensitive manner.
British Educational Research Association (2018) Ethical Guidelines for Educational Research. Available at: https://www.bera.ac.uk/publication/ethical-guidelines-for-educational-research-2018 (Accessed: 9 June 2023).
British Psychological Society (2021) Code of Human Research Ethics . Available at: https://cms.bps.org.uk/sites/default/files/2022-06/BPS%20Code%20of%20Human%20Research%20Ethics%20%281%29.pdf (Accessed: 9 June 2023).
British Sociological Association (2016) Researching online forums . Available at: https://www.britsoc.co.uk/media/24834/j000208_researching_online_forums_-cs1-_v3.pdf (Accessed: 9 June 2023).
Health Research Authority (2022) UK Policy Framework for Health and Social Care Research . Available at: https://www.hra.nhs.uk/planning-and-improving-research/policies-standards-legislation/uk-policy-framework-health-social-care-research/uk-policy-framework-health-and-social-care-research/#chiefinvestigators (Accessed: 9 June 2023).
Heslop, C., Burns, S., Lobo, R. (2018) ‘Managing qualitative research as insider-research in small rural communities’, Rural and Remote Health , 18: pp. 4576.
Equality Act 2010, c. 15. Available at: https://www.legislation.gov.uk/ukpga/2010/15/introduction (Accessed: 9 June 2023).
National Institute for Health and Care Research (no date) Equality, Diversity and Inclusion (EDI) . Available at: https://arc-kss.nihr.ac.uk/public-and-community-involvement/pcie-guide/how-to-do-pcie/equality-diversity-and-inclusion-edi (Accessed: 9 June 2023).
National Institute for Health and Care Research (2020) Improving inclusion of under-served groups in clinical research: Guidance from INCLUDE project. Available at: https://www.nihr.ac.uk/documents/improving-inclusion-of-under-served-groups-in-clinical-research-guidance-from-include-project/25435 (Accessed: 9 June 2023).
Sapiro, B. and Matthews, E. (2020) ‘Both Insider and Outsider. On Conducting Social Work Research in Mental Health Settings’, Advances in Social Work , 20(3). Available at: https://doi.org/10.18060/23926
Turcotte-Tremblay, A. and McSween-Cadieux, E. (2018) ‘A reflection on the challenge of protecting confidentiality of participants when disseminating research results locally’, BMC Medical Ethics, 19(supplement 1), no. 45. Available at: https://bmcmedethics.biomedcentral.com/articles/10.1186/s12910-018-0279-0
United Nations General Assembly (1948) The Universal Declaration of Human Rights . Resolution A/RES/217/A. Available at: https://www.un.org/en/about-us/universal-declaration-of-human-rights#:~:text=Drafted%20by%20representatives%20with%20different,all%20peoples%20and%20all%20nations . (Accessed: 9 June 2023).
Wellcome Trust (2014) Ensuring your research is ethical: A guide for Extended Project Qualification students . Available at: https://wellcome.org/sites/default/files/wtp057673_0.pdf (Accessed: 9 June 2023).
More articles from the research proposal collection

Writing your research proposal
A doctoral research degree is the highest academic qualification that a student can achieve. The guidance provided in these articles will help you apply for one of the two main types of research degree offered by The Open University.
Level: 1 Introductory

Defining your research methodology
Your research methodology is the approach you will take to guide your research process and explain why you use particular methods. This article explains more.

Writing your proposal and preparing for your interview
The final article looks at writing your research proposal - from the introduction through to citations and referencing - as well as preparing for your interview.
Free courses on postgraduate study

Are you ready for postgraduate study?
This free course, Are you ready for postgraduate study, will help you to become familiar with the requirements and demands of postgraduate study and ensure you are ready to develop the skills and confidence to pursue your learning further.

Succeeding in postgraduate study
This free course, Succeeding in postgraduate study, will help you to become familiar with the requirements and demands of postgraduate study and to develop the skills and confidence to pursue your learning further.

Applying to study for a PhD in psychology
This free OpenLearn course is for psychology students and graduates who are interested in PhD study at some future point. Even if you have met PhD students and heard about their projects, it is likely that you have only a vague idea of what PhD study entails. This course is intended to give you more information.
Become an OU student
Ratings & comments, share this free course, copyright information, publication details.
- Originally published: Tuesday, 27 June 2023
- Body text - Creative Commons BY-NC-SA 4.0 : The Open University
- Image 'Pebbles balance on a stone see-saw' - Copyright: Photo 51106733 / Balance © Anatoli Styf | Dreamstime.com
- Image 'Camera equipment set up filming a man talking' - Copyright: Photo 42631221 © Gabriel Robledo | Dreamstime.com
- Image 'Applying to study for a PhD in psychology' - Copyright free
- Image 'Succeeding in postgraduate study' - Copyright: © Everste/Getty Images
- Image 'Addressing ethical issues in your research proposal' - Copyright: Photo 50384175 / Children Playing © Lenutaidi | Dreamstime.com
- Image 'Writing your proposal and preparing for your interview' - Copyright: Photo 133038259 / Black Student © Fizkes | Dreamstime.com
- Image 'Defining your research methodology' - Copyright free
- Image 'Writing your research proposal' - Copyright free
- Image 'Are you ready for postgraduate study?' - Copyright free
Rate and Review
Rate this article, review this article.
Log into OpenLearn to leave reviews and join in the conversation.
Article reviews
For further information, take a look at our frequently asked questions which may give you the support you need.
- Privacy Policy

Home » Ethical Considerations – Types, Examples and Writing Guide
Ethical Considerations – Types, Examples and Writing Guide
Table of Contents

Ethical Considerations
Ethical considerations in research refer to the principles and guidelines that researchers must follow to ensure that their studies are conducted in an ethical and responsible manner. These considerations are designed to protect the rights, safety, and well-being of research participants, as well as the integrity and credibility of the research itself
Some of the key ethical considerations in research include:
- Informed consent: Researchers must obtain informed consent from study participants, which means they must inform participants about the study’s purpose, procedures, risks, benefits, and their right to withdraw at any time.
- Privacy and confidentiality : Researchers must ensure that participants’ privacy and confidentiality are protected. This means that personal information should be kept confidential and not shared without the participant’s consent.
- Harm reduction : Researchers must ensure that the study does not harm the participants physically or psychologically. They must take steps to minimize the risks associated with the study.
- Fairness and equity : Researchers must ensure that the study does not discriminate against any particular group or individual. They should treat all participants equally and fairly.
- Use of deception: Researchers must use deception only if it is necessary to achieve the study’s objectives. They must inform participants of the deception as soon as possible.
- Use of vulnerable populations : Researchers must be especially cautious when working with vulnerable populations, such as children, pregnant women, prisoners, and individuals with cognitive or intellectual disabilities.
- Conflict of interest : Researchers must disclose any potential conflicts of interest that may affect the study’s integrity. This includes financial or personal relationships that could influence the study’s results.
- Data manipulation: Researchers must not manipulate data to support a particular hypothesis or agenda. They should report the results of the study objectively, even if the findings are not consistent with their expectations.
- Intellectual property: Researchers must respect intellectual property rights and give credit to previous studies and research.
- Cultural sensitivity : Researchers must be sensitive to the cultural norms and beliefs of the participants. They should avoid imposing their values and beliefs on the participants and should be respectful of their cultural practices.
Types of Ethical Considerations
Types of Ethical Considerations are as follows:
Research Ethics:
This includes ethical principles and guidelines that govern research involving human or animal subjects, ensuring that the research is conducted in an ethical and responsible manner.
Business Ethics :
This refers to ethical principles and standards that guide business practices and decision-making, such as transparency, honesty, fairness, and social responsibility.
Medical Ethics :
This refers to ethical principles and standards that govern the practice of medicine, including the duty to protect patient autonomy, informed consent, confidentiality, and non-maleficence.
Environmental Ethics :
This involves ethical principles and values that guide our interactions with the natural world, including the obligation to protect the environment, minimize harm, and promote sustainability.
Legal Ethics
This involves ethical principles and standards that guide the conduct of legal professionals, including issues such as confidentiality, conflicts of interest, and professional competence.
Social Ethics
This involves ethical principles and values that guide our interactions with other individuals and society as a whole, including issues such as justice, fairness, and human rights.
Information Ethics
This involves ethical principles and values that govern the use and dissemination of information, including issues such as privacy, accuracy, and intellectual property.
Cultural Ethics
This involves ethical principles and values that govern the relationship between different cultures and communities, including issues such as respect for diversity, cultural sensitivity, and inclusivity.
Technological Ethics
This refers to ethical principles and guidelines that govern the development, use, and impact of technology, including issues such as privacy, security, and social responsibility.
Journalism Ethics
This involves ethical principles and standards that guide the practice of journalism, including issues such as accuracy, fairness, and the public interest.
Educational Ethics
This refers to ethical principles and standards that guide the practice of education, including issues such as academic integrity, fairness, and respect for diversity.
Political Ethics
This involves ethical principles and values that guide political decision-making and behavior, including issues such as accountability, transparency, and the protection of civil liberties.
Professional Ethics
This refers to ethical principles and standards that guide the conduct of professionals in various fields, including issues such as honesty, integrity, and competence.
Personal Ethics
This involves ethical principles and values that guide individual behavior and decision-making, including issues such as personal responsibility, honesty, and respect for others.
Global Ethics
This involves ethical principles and values that guide our interactions with other nations and the global community, including issues such as human rights, environmental protection, and social justice.
Applications of Ethical Considerations
Ethical considerations are important in many areas of society, including medicine, business, law, and technology. Here are some specific applications of ethical considerations:
- Medical research : Ethical considerations are crucial in medical research, particularly when human subjects are involved. Researchers must ensure that their studies are conducted in a way that does not harm participants and that participants give informed consent before participating.
- Business practices: Ethical considerations are also important in business, where companies must make decisions that are socially responsible and avoid activities that are harmful to society. For example, companies must ensure that their products are safe for consumers and that they do not engage in exploitative labor practices.
- Environmental protection: Ethical considerations play a crucial role in environmental protection, as companies and governments must weigh the benefits of economic development against the potential harm to the environment. Decisions about land use, resource allocation, and pollution must be made in an ethical manner that takes into account the long-term consequences for the planet and future generations.
- Technology development : As technology continues to advance rapidly, ethical considerations become increasingly important in areas such as artificial intelligence, robotics, and genetic engineering. Developers must ensure that their creations do not harm humans or the environment and that they are developed in a way that is fair and equitable.
- Legal system : The legal system relies on ethical considerations to ensure that justice is served and that individuals are treated fairly. Lawyers and judges must abide by ethical standards to maintain the integrity of the legal system and to protect the rights of all individuals involved.
Examples of Ethical Considerations
Here are a few examples of ethical considerations in different contexts:
- In healthcare : A doctor must ensure that they provide the best possible care to their patients and avoid causing them harm. They must respect the autonomy of their patients, and obtain informed consent before administering any treatment or procedure. They must also ensure that they maintain patient confidentiality and avoid any conflicts of interest.
- In the workplace: An employer must ensure that they treat their employees fairly and with respect, provide them with a safe working environment, and pay them a fair wage. They must also avoid any discrimination based on race, gender, religion, or any other characteristic protected by law.
- In the media : Journalists must ensure that they report the news accurately and without bias. They must respect the privacy of individuals and avoid causing harm or distress. They must also be transparent about their sources and avoid any conflicts of interest.
- In research: Researchers must ensure that they conduct their studies ethically and with integrity. They must obtain informed consent from participants, protect their privacy, and avoid any harm or discomfort. They must also ensure that their findings are reported accurately and without bias.
- In personal relationships : People must ensure that they treat others with respect and kindness, and avoid causing harm or distress. They must respect the autonomy of others and avoid any actions that would be considered unethical, such as lying or cheating. They must also respect the confidentiality of others and maintain their privacy.
How to Write Ethical Considerations
When writing about research involving human subjects or animals, it is essential to include ethical considerations to ensure that the study is conducted in a manner that is morally responsible and in accordance with professional standards. Here are some steps to help you write ethical considerations:
- Describe the ethical principles: Start by explaining the ethical principles that will guide the research. These could include principles such as respect for persons, beneficence, and justice.
- Discuss informed consent : Informed consent is a critical ethical consideration when conducting research. Explain how you will obtain informed consent from participants, including how you will explain the purpose of the study, potential risks and benefits, and how you will protect their privacy.
- Address confidentiality : Describe how you will protect the confidentiality of the participants’ personal information and data, including any measures you will take to ensure that the data is kept secure and confidential.
- Consider potential risks and benefits : Describe any potential risks or harms to participants that could result from the study and how you will minimize those risks. Also, discuss the potential benefits of the study, both to the participants and to society.
- Discuss the use of animals : If the research involves the use of animals, address the ethical considerations related to animal welfare. Explain how you will minimize any potential harm to the animals and ensure that they are treated ethically.
- Mention the ethical approval : Finally, it’s essential to acknowledge that the research has received ethical approval from the relevant institutional review board or ethics committee. State the name of the committee, the date of approval, and any specific conditions or requirements that were imposed.
When to Write Ethical Considerations
Ethical considerations should be written whenever research involves human subjects or has the potential to impact human beings, animals, or the environment in some way. Ethical considerations are also important when research involves sensitive topics, such as mental health, sexuality, or religion.
In general, ethical considerations should be an integral part of any research project, regardless of the field or subject matter. This means that they should be considered at every stage of the research process, from the initial planning and design phase to data collection, analysis, and dissemination.
Ethical considerations should also be written in accordance with the guidelines and standards set by the relevant regulatory bodies and professional associations. These guidelines may vary depending on the discipline, so it is important to be familiar with the specific requirements of your field.
Purpose of Ethical Considerations
Ethical considerations are an essential aspect of many areas of life, including business, healthcare, research, and social interactions. The primary purposes of ethical considerations are:
- Protection of human rights: Ethical considerations help ensure that people’s rights are respected and protected. This includes respecting their autonomy, ensuring their privacy is respected, and ensuring that they are not subjected to harm or exploitation.
- Promoting fairness and justice: Ethical considerations help ensure that people are treated fairly and justly, without discrimination or bias. This includes ensuring that everyone has equal access to resources and opportunities, and that decisions are made based on merit rather than personal biases or prejudices.
- Promoting honesty and transparency : Ethical considerations help ensure that people are truthful and transparent in their actions and decisions. This includes being open and honest about conflicts of interest, disclosing potential risks, and communicating clearly with others.
- Maintaining public trust: Ethical considerations help maintain public trust in institutions and individuals. This is important for building and maintaining relationships with customers, patients, colleagues, and other stakeholders.
- Ensuring responsible conduct: Ethical considerations help ensure that people act responsibly and are accountable for their actions. This includes adhering to professional standards and codes of conduct, following laws and regulations, and avoiding behaviors that could harm others or damage the environment.
Advantages of Ethical Considerations
Here are some of the advantages of ethical considerations:
- Builds Trust : When individuals or organizations follow ethical considerations, it creates a sense of trust among stakeholders, including customers, clients, and employees. This trust can lead to stronger relationships and long-term loyalty.
- Reputation and Brand Image : Ethical considerations are often linked to a company’s brand image and reputation. By following ethical practices, a company can establish a positive image and reputation that can enhance its brand value.
- Avoids Legal Issues: Ethical considerations can help individuals and organizations avoid legal issues and penalties. By adhering to ethical principles, companies can reduce the risk of facing lawsuits, regulatory investigations, and fines.
- Increases Employee Retention and Motivation: Employees tend to be more satisfied and motivated when they work for an organization that values ethics. Companies that prioritize ethical considerations tend to have higher employee retention rates, leading to lower recruitment costs.
- Enhances Decision-making: Ethical considerations help individuals and organizations make better decisions. By considering the ethical implications of their actions, decision-makers can evaluate the potential consequences and choose the best course of action.
- Positive Impact on Society: Ethical considerations have a positive impact on society as a whole. By following ethical practices, companies can contribute to social and environmental causes, leading to a more sustainable and equitable society.
About the author
Muhammad Hassan
Researcher, Academic Writer, Web developer
You may also like

How to Cite Research Paper – All Formats and...

Data Collection – Methods Types and Examples

Delimitations in Research – Types, Examples and...

Research Paper Format – Types, Examples and...

Research Process – Steps, Examples and Tips

Research Design – Types, Methods and Examples
Ethical considerations in research: Best practices and examples

To conduct responsible research, you’ve got to think about ethics. They protect participants’ rights and their well-being - and they ensure your findings are valid and reliable. This isn’t just a box for you to tick. It’s a crucial consideration that can make all the difference to the outcome of your research.
In this article, we'll explore the meaning and importance of research ethics in today's research landscape. You'll learn best practices to conduct ethical and impactful research.
Examples of ethical considerations in research
As a researcher, you're responsible for ethical research alongside your organization. Fulfilling ethical guidelines is critical. Organizations must ensure employees follow best practices to protect participants' rights and well-being.
Keep these things in mind when it comes to ethical considerations in research:
Voluntary participation
Voluntary participation is key. Nobody should feel like they're being forced to participate or pressured into doing anything they don't want to. That means giving people a choice and the ability to opt out at any time, even if they've already agreed to take part in the study.
Informed consent
Informed consent isn't just an ethical consideration. It's a legal requirement as well. Participants must fully understand what they're agreeing to, including potential risks and benefits.
The best way to go about this is by using a consent form. Make sure you include:
- A brief description of the study and research methods.
- The potential benefits and risks of participating.
- The length of the study.
- Contact information for the researcher and/or sponsor.
- Reiteration of the participant’s right to withdraw from the research project at any time without penalty.
Anonymity means that participants aren't identifiable in any way. This includes:
- Email address
- Photographs
- Video footage
You need a way to anonymize research data so that it can't be traced back to individual participants. This may involve creating a new digital ID for participants that can’t be linked back to their original identity using numerical codes.
Confidentiality
Information gathered during a study must be kept confidential. Confidentiality helps to protect the privacy of research participants. It also ensures that their information isn't disclosed to unauthorized individuals.
Some ways to ensure confidentiality include:
- Using a secure server to store data.
- Removing identifying information from databases that contain sensitive data.
- Using a third-party company to process and manage research participant data.
- Not keeping participant records for longer than necessary.
- Avoiding discussion of research findings in public forums.
Potential for harm
The potential for harm is a crucial factor in deciding whether a research study should proceed. It can manifest in various forms, such as:
- Psychological harm
- Social harm
- Physical harm
Conduct an ethical review to identify possible harms. Be prepared to explain how you’ll minimize these harms and what support is available in case they do happen.
Fair payment
One of the most crucial aspects of setting up a research study is deciding on fair compensation for your participants. Underpayment is a common ethical issue that shouldn't be overlooked. Properly rewarding participants' time is critical for boosting engagement and obtaining high-quality data. While Prolific requires a minimum payment of £6.00 / $8.00 per hour, there are other factors you need to consider when deciding on a fair payment.
First, check your institution's reimbursement guidelines to see if they already have a minimum or maximum hourly rate. You can also use the national minimum wage as a reference point.
Next, think about the amount of work you're asking participants to do. The level of effort required for a task, such as producing a video recording versus a short survey, should correspond with the reward offered.
You also need to consider the population you're targeting. To attract research subjects with specific characteristics or high-paying jobs, you may need to offer more as an incentive.
We recommend a minimum payment of £9.00 / $12.00 per hour, but we understand that payment rates can vary depending on a range of factors. Whatever payment you choose should reflect the amount of effort participants are required to put in and be fair to everyone involved.
Ethical research made easy with Prolific
At Prolific, we believe in making ethical research easy and accessible. The findings from the Fairwork Cloudwork report speak for themselves. Prolific was given the top score out of all competitors for minimum standards of fair work.
With over 25,000 researchers in our community, we're leading the way in revolutionizing the research industry. If you're interested in learning more about how we can support your research journey, sign up to get started now.
You might also like

High-quality human data to deliver world-leading research and AIs.

Follow us on
All Rights Reserved Prolific 2024

What Are the Ethical Considerations in Research Design?
When I began my work on the thesis I was always focused on my research. However, once I began to make my way through research, I realized that research ethics is a core aspect of the research work and the foundation of research design.
Research ethics play a crucial role in ensuring the responsible conduct of research. Here are some key reasons why research ethics matter:

Let us look into some of the major ethical considerations in research design.
Ethical Issues in Research
There are many organizations, like the Committee on Publication Ethics , dedicated to promoting ethics in scientific research. These organizations agree that ethics is not an afterthought or side note to the research study. It is an integral aspect of research that needs to remain at the forefront of our work.
The research design must address specific research questions. Hence, the conclusions of the study must correlate to the questions posed and the results. Also, research ethics demands that the methods used must relate specifically to the research questions.
Voluntary Participation and Consent
An individual should at no point feel any coercion to participate in a study. This includes any type of persuasion or deception in attempting to gain an individual’s trust.
Informed consent states that an individual must give their explicit consent to participate in the study. You can think of consent form as an agreement of trust between the researcher and the participants.
Sampling is the first step in research design . You will need to explain why you want a particular group of participants. You will have to explain why you left out certain people or groups. In addition, if your sample includes children or special needs individuals, you will have additional requirements to address like parental permission.
Confidentiality
The third ethics principle of the Economic and Social Research Council (ESRC) states that: “The confidentiality of the information supplied by research subjects and the anonymity of respondents must be respected.” However, sometimes confidentiality is limited. For example, if a participant is at risk of harm, we must protect them. This might require releasing confidential information.
Risk of Harm
We should do everything in our power to protect study participants. For this, we should focus on the risk to benefit ratio. If possible risks outweigh the benefits, then we should abandon or redesign the study. Risk of harm also requires us to measure the risk to benefit ratio as the study progresses.
Research Methods
We know there are numerous research methods. However, when it comes to ethical considerations, some key questions can help us find the right approach for our studies.
i. Which methods most effectively fit the aims of your research?
ii. What are the strengths and restrictions of a particular method?
iii. Are there potential risks when using a particular research method?
For more guidance, you can refer to the ESRC Framework for Research Ethics .
Ethical issues in research can arise at various stages of the research process and involve different aspects of the study. Here are some common examples of ethical issues in research:
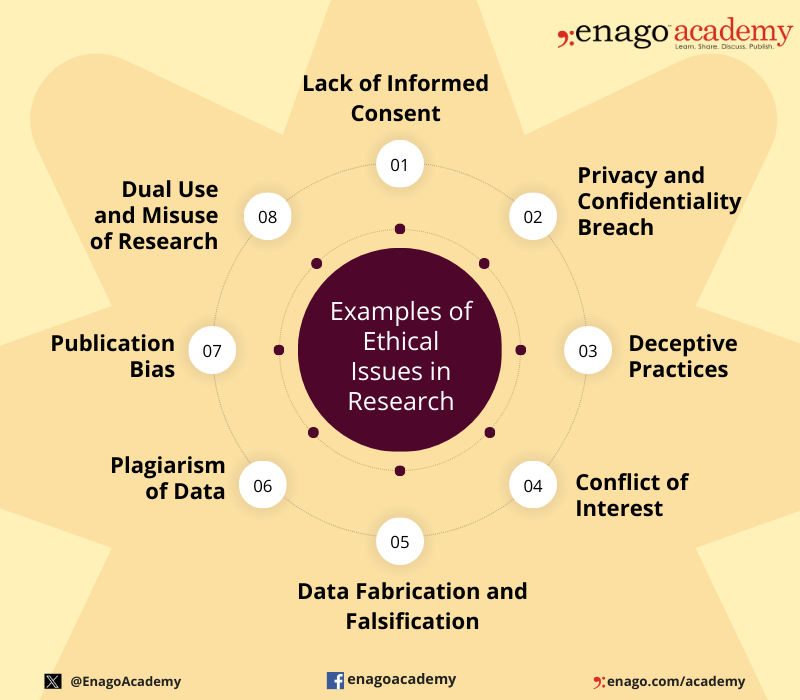
Institutional Review Boards
The importance of ethics in research cannot be understated. Following ethical guidelines will ensure your study’s validity and promote its contribution to scientific study. On a personal level, you will strengthen your research and increase your opportunities to gain funding.
To address the need for ethical considerations, most institutions have their own Institutional Review Board (IRB). An IRB secures the safety of human participants and prevents violation of human rights. It reviews the research aims and methodologies to ensure ethical practices are followed. If a research design does not follow the set ethical guidelines, then the researcher will have to amend their study.
Applying for Ethical Approval
Applications for ethical approval will differ across institutions. Regardless, they focus on the benefits of your research and the risk to benefit ratio concerning participants. Therefore, you need to effectively address both in order to get ethical clearence.
Participants
It is vital that you make it clear that individuals are provided with sufficient information in order to make an informed decision on their participation. In addition, you need to demonstrate that the ethical issues of consent, risk of harm, and confidentiality are clearly defined.
Benefits of the Study
You need to prove to the panel that your work is essential and will yield results that contribute to the scientific community. For this, you should demonstrate the following:
i. The conduct of research guarantees the quality and integrity of results.
ii. The research will be properly distributed.
iii. The aims of the research are clear and the methodology is appropriate.
Integrity and transparency are vital in the research. Ethics committees expect you to share any actual or potential conflicts of interest that could affect your work. In addition, you have to be honest and transparent throughout the approval process and the research process.
The Dangers of Unethical Practices
There is a reason to follow ethical guidelines. Without these guidelines, our research will suffer. Moreover, more importantly, people could suffer.
The following are just two examples of infamous cases of unethical research practices that demonstrate the importance of adhering to ethical standards:
- The Stanford Prison Experiment (1971) aimed to investigate the psychological effects of power using the relationship between prisoners and prison officers. Those assigned the role of “prison officers” embraced measures that exposed “prisoners” to psychological and physical harm. In this case, there was voluntary participation. However, there was disregard for welfare of the participants.
- Recently, Chinese scientist He Jiankui announced his work on genetically edited babies . Over 100 Chinese scientists denounced this research, calling it “crazy” and “shocking and unacceptable.” This research shows a troubling attitude of “do first, debate later” and a disregard for the ethical concerns of manipulating the human body Wang Yuedan, a professor of immunology at Peking University, calls this “an ethics disaster for the world” and demands strict punishments for this type of ethics violation.
What are your experiences with research ethics? How have you developed an ethical approach to research design? Please share your thoughts with us in the comments section below.
I love the articulation of reasoning and practical examples of unethical research
Rate this article Cancel Reply
Your email address will not be published.

Enago Academy's Most Popular Articles

- AI in Academia
- Trending Now
6 Leading AI Detection Tools for Academic Writing — A comparative analysis
The advent of AI content generators, exemplified by advanced models like ChatGPT, Claude AI, and…

- Reporting Research
Choosing the Right Analytical Approach: Thematic analysis vs. content analysis for data interpretation
In research, choosing the right approach to understand data is crucial for deriving meaningful insights.…

- Industry News
China’s Ministry of Education Spearheads Efforts to Uphold Academic Integrity
In response to the increase in retractions of research papers submitted by Chinese scholars to…

Comparing Cross Sectional and Longitudinal Studies: 5 steps for choosing the right approach
The process of choosing the right research design can put ourselves at the crossroads of…

- Publishing Research
- Understanding Ethics
Understanding the Difference Between Research Ethics and Compliance
Ethics refers to the principles, values, and moral guidelines that guide individual or group behavior…
Unlocking the Power of Networking in Academic Conferences
Intersectionality in Academia: Dealing with diverse perspectives
Meritocracy and Diversity in Science: Increasing inclusivity in STEM education
Avoiding the AI Trap: Pitfalls of relying on ChatGPT for PhD applications

Sign-up to read more
Subscribe for free to get unrestricted access to all our resources on research writing and academic publishing including:
- 2000+ blog articles
- 50+ Webinars
- 10+ Expert podcasts
- 50+ Infographics
- 10+ Checklists
- Research Guides
We hate spam too. We promise to protect your privacy and never spam you.
I am looking for Editing/ Proofreading services for my manuscript Tentative date of next journal submission:

As a researcher, what do you consider most when choosing an image manipulation detector?
A guide to ethical considerations in research
Last updated
12 March 2023
Reviewed by
Miroslav Damyanov
Whether you are conducting a survey, running focus groups , doing field research, or holding interviews, the chances are participants will be a part of the process.
Taking ethical considerations into account and following all obligations are essential when people are involved in your research. Upholding academic integrity is another crucial ethical concern in all research types.
So, how can you protect your participants and ensure that your research is ethical? Let’s take a closer look at the ethical considerations in research and the best practices to follow.
Make research less tedious
Dovetail streamlines research to help you uncover and share actionable insights
- The importance of ethical research
Research ethics are integral to all forms of research. They help protect participants’ rights, ensure that the research is valid and accurate, and help minimize any risk of harm during the process.
When people are involved in your research, it’s particularly important to consider whether your planned research method follows ethical practices.
You might ask questions such as:
Will our participants be protected?
Is there a risk of any harm?
Are we doing all we can to protect the personal data and information we collect?
Does our study include any bias?
How can we ensure that the results will be accurate and valid?
Will our research impact public safety?
Is there a more ethical way to complete the research?
Conducting research unethically and not protecting participants’ rights can have serious consequences. It can discredit the entire study. Human rights, dignity, and research integrity should all be front of mind when you are conducting research.
- How to conduct ethical research
Before kicking off any project, the entire team must be familiar with ethical best practices. These include the considerations below.
Voluntary participation
In an ethical study, all participants have chosen to be part of the research. They must have voluntarily opted in without any pressure or coercion to do so. They must be aware that they are part of a research study. Their information must not be used against their will.
To ensure voluntary participation, make it clear at the outset that the person is opting into the process.
While participants may agree to be part of a study for a certain duration, they are allowed to change their minds. Participants must be free to leave or withdraw from the study at any time. They don’t need to give a reason.
Informed consent
Before kicking off any research, it’s also important to gain consent from all participants. This ensures participants are clear that they are part of a research study and understand all of the information related to it.
Gaining informed consent usually involves a written consent form—physical or digital—that participants can sign.
Best practice informed consent generally includes the following:
An explanation of what the study is
The duration of the study
The expectations of participants
Any potential risks
An explanation that participants are free to withdraw at any time
Contact information for the research supervisor
When obtaining informed consent, you should ensure that all parties truly understand what they are signing and their obligations as a participant. There should never be any coercion to sign.
Anonymity is key to ensuring that participants cannot be identified through their data. Personal information includes things like participants’ names, addresses, emails, phone numbers, characteristics, and photos.
However, making information truly anonymous can be challenging, especially if personal information is a necessary part of the research.
To maintain a degree of anonymity, avoid gathering any information you don’t need. This will minimize the risk of participants being identified.
Another useful tool is data pseudonymization, which makes it harder to directly link information to a real person. Data pseudonymization means giving participants fake names or mock information to protect their identity. You could, for example, replace participants’ names with codes.
Confidentiality
Keeping data confidential is a critical aspect of all forms of research. You should communicate to all participants that their information will be protected and then take active steps to ensure that happens.
Data protection has become a serious topic in recent years and should be taken seriously. The more information you gather, the more important it is to heavily protect that data.
There are many ways to protect data, including the following:
Restricted access: Information should only be accessible to the researchers involved in the project to limit the risk of breaches.
Password protection : Information should not be accessible without access via a password that complies with secure password guidelines.
Encrypted data: In this day and age, password protection isn’t usually sufficient. Encrypting the data can help ensure its security.
Data retention: All organizations should uphold a data retention policy whereby data gathered should only be held for a certain period of time. This minimizes the risk of breaches further down the line.
In research where participants are grouped together (such as in focus groups), ask participants not to pass on what has been discussed. This helps maintain the group’s privacy.
Data falsification
Regardless of what your study is about or whether it involves humans, it’s always unethical to falsify data or information. That means editing or changing any data that has been gathered or gathering data in ways that skew the results.
Bias in research is highly problematic and can significantly impact research integrity. Data falsification or misrepresentation can have serious consequences.
Take the case of Korean researcher Hwang Woo-suk, for example. Woo-suk, once considered a scientific leader in stem-cell research, was found guilty of fabricating experiments in the field and making ethical violations. Once discovered, he was fired from his role and sentenced to two years in prison.
All conflicts of interest should be declared at the outset to avoid any bias or risk of fabrication in the research process. Data must be collected and recorded accurately, and analysis must be completed impartially.
If conflicts do arise during the study, researchers may need to step back to maintain the study’s integrity. Outsourcing research to neutral third parties is necessary in some cases.
Potential for harm
Another consideration is the potential for harm. When completing research, it’s important to ensure that your participants will be safe throughout the study’s duration.
Harm during research could occur in many forms.
Physical harm may occur if your participants are asked to perform a physical activity, or if they are involved in a medical study.
Psychological harm can occur if questions or activities involve triggering or sensitive topics, or if participants are asked to complete potentially embarrassing tasks.
Harm can be caused through a data breach or privacy concern.
A study can cause harm if the participants don’t feel comfortable with the study expectations or their supervisors.
Maintaining the physical and mental well-being of all participants throughout studies is an essential aspect of ethical research.
- Gaining ethical approval
Gaining ethical approval may be necessary before conducting some types of research.
The US Department of Health and Human Services (HHS) and the US Food and Drug Administration (FDA) advise that approval is likely required for studies involving people.
To gain approval, it’s necessary to submit a proposal to an Institutional Review Board (IRB). The board will check the proposal and ensure that the research aligns with ethical practices. It will allow the project to proceed if it meets requirements.
Not gaining appropriate approval could invalidate your study, so it’s essential to pay attention to all local guidelines and laws.
- The dangers of unethical practices
Not maintaining ethical standards in research isn’t just questionable—it can be dangerous too. Many historical cases show just how widespread the ramifications can be.
The case of Korean researcher Hwang Woo-suk shows just how critical it is to obtain information ethically and accurately represent findings.
A case in 1998, which involved fraudulent data reporting, further proves this point.
The study, now debunked, was completed by Andrew Wakefield. It suggested there may be a link between the measles, mumps, and rubella (MMR) vaccine and autism in children. It was later found that the data was manipulated to show a causal link when there wasn’t one. Wakefield’s medical license was removed as a result, but the fraudulent study was still widely cited and continues to cause vaccine hesitancy among many parents.
Large organizational bodies have also been a part of unethical research. The alcohol industry, for example, was found to be highly influential in a major public health study in an attempt to prove that moderate alcohol consumption had health benefits. Five major alcohol companies pledged approximately $66 million to fund the study.
However, the World Health Organization (WHO) is clear that research shows there is no safe level of alcohol consumption. After pressure from many organizations, the study was eventually pulled due to biasing by the alcohol industry. Despite this, the idea that moderate alcohol consumption is better than abstaining may still appear in public discourse.
In more extreme cases, unethical research has led to medical studies being completed on people without their knowledge and against their will. The atrocities committed in Nazi Germany during World War II are an example.
Unethical practices in research are not just problematic or in conflict with academic integrity; they can seriously harm public health and safety.
- The ethical way to research
Considering ethical concerns and adopting best practices throughout studies is essential when conducting research.
When people are involved in studies, it’s important to consider their rights. They must not be coerced into participating, and they should be protected throughout the process.
Accurate reporting, unbiased results, and a genuine interest in answering questions rather than confirming assumptions are all essential aspects of ethical research.
Ethical research ultimately means producing true and valuable results for the benefit of everyone impacted by your study.
What are ethical considerations in research?
Ethical research involves a series of guidelines and considerations to ensure that the information gathered is valid and reliable. These guidelines ensure that:
People are not harmed during research
Participants have data protection and anonymity
Academic integrity is upheld
Not maintaining ethics in research can have serious consequences for those involved in the studies, the broader public, and policymakers.
What are the most common ethical considerations?
To maintain integrity and validity in research, all biases must be removed, data should be reported accurately, and studies must be clearly represented.
Some of the most common ethical guidelines when it comes to humans in research include avoiding harm, data protection, anonymity, informed consent, and confidentiality.

What are the ethical issues in secondary research?
Using secondary data is generally considered an ethical practice. That’s because the use of secondary data minimizes the impact on participants, reduces the need for additional funding, and maximizes the value of the data collection.
However, secondary research still has risks. For example, the risk of data breaches increases as more parties gain access to the information.
To minimize the risk, researchers should consider anonymity or data pseudonymization before the data is passed on. Furthermore, using the data should not cause any harm or distress to participants.
Should you be using a customer insights hub?
Do you want to discover previous research faster?
Do you share your research findings with others?
Do you analyze research data?
Start for free today, add your research, and get to key insights faster
Editor’s picks
Last updated: 11 January 2024
Last updated: 15 January 2024
Last updated: 17 January 2024
Last updated: 12 May 2023
Last updated: 30 April 2024
Last updated: 18 May 2023
Last updated: 25 November 2023
Last updated: 13 May 2024
Latest articles
Related topics, .css-je19u9{-webkit-align-items:flex-end;-webkit-box-align:flex-end;-ms-flex-align:flex-end;align-items:flex-end;display:-webkit-box;display:-webkit-flex;display:-ms-flexbox;display:flex;-webkit-flex-direction:row;-ms-flex-direction:row;flex-direction:row;-webkit-box-flex-wrap:wrap;-webkit-flex-wrap:wrap;-ms-flex-wrap:wrap;flex-wrap:wrap;-webkit-box-pack:center;-ms-flex-pack:center;-webkit-justify-content:center;justify-content:center;row-gap:0;text-align:center;max-width:671px;}@media (max-width: 1079px){.css-je19u9{max-width:400px;}.css-je19u9>span{white-space:pre;}}@media (max-width: 799px){.css-je19u9{max-width:400px;}.css-je19u9>span{white-space:pre;}} decide what to .css-1kiodld{max-height:56px;display:-webkit-box;display:-webkit-flex;display:-ms-flexbox;display:flex;-webkit-align-items:center;-webkit-box-align:center;-ms-flex-align:center;align-items:center;}@media (max-width: 1079px){.css-1kiodld{display:none;}} build next, decide what to build next.

Users report unexpectedly high data usage, especially during streaming sessions.

Users find it hard to navigate from the home page to relevant playlists in the app.

It would be great to have a sleep timer feature, especially for bedtime listening.

I need better filters to find the songs or artists I’m looking for.
Log in or sign up
Get started for free
Ethical Issues in Research
- Living reference work entry
- First Online: 05 March 2021
- Cite this living reference work entry

- Juwel Rana 2 , 3 , 4 ,
- Segufta Dilshad 2 &
- Md. Ali Ahsan 5
365 Accesses
2 Citations
The most important human endeavor is the striving for morality in our actions. Our inner balance and even our very existence depend on it. Only morality in our actions can give beauty and dignity to life – Albert Einstein.
Ethics ; Methodology ; Mixed-method research ; Observation ; Qualitative research ; Quantitative research ; Research ; Research design ; Research ethics
Ethics is a set of standards, a code, or value system, worked out from human reason and experience, by which free human actions are determined as ultimately right or wrong, good, or evil. If acting agrees with these standards, it is ethical, otherwise unethical.
Scientific research refers to a persistent exercise towards producing new knowledge to unveil a new stream of ideas in academia for humankind.
Research ethics refer to some of the genres that researchers follow to protect the rights in developing research strategies and building a trusted relationship between the...
This is a preview of subscription content, log in via an institution to check access.
Access this chapter
Institutional subscriptions
Bulmer M (1982) Social Research Ethics: An Examination of the Merits of Covert Participant Observation. Holmes & Meier Publishers
Google Scholar
Butler I (2002) A Code of Ethics for Social Work and Social Care Research. Br J Soc Work [Internet]. 32(2):239–48. Available from: https://doi.org/10.1093/bjsw/32.2.239
Fisher CB, Anushko AE (2008) The SAGE Handbook of Social Research Methods [Internet]. London: SAGE Publications Ltd; p. 95–109. Available from: https://methods.sagepub.com/book/the-sage-handbook-of-socialresearch-methods
Hill J, Wright LT (2001) A qualitative research agenda for small to medium-sized enterprises. Mark Intell Plan 19(6):432–443
Homan R (1991) The ethics of social research. Addison-Wesley Longman Limited
Israel M, Hay I (2006) Research ethics for social scientists. Sage
Kimmel AJ (1988) Ethics and values in applied social research. 1st ed. SAGE Publications Inc
Orb A, Eisenhauer L, Wynaden D (2001) Ethics in qualitative research. J Nurs Scholarsh 33(1):93–96
Principles of research ethics [Internet]. Lund Research Ltd. 2012 [cited 2020 Dec 15]. Available from: https://dissertation.laerd.com/principles-of-research-ethics.php
Robley LR (1995) The ethics of qualitative nursing research. J Prof Nurs 11(1):45–48
Wiles R, Charles V, Crow G, Heath S (2006) Researching researchers: lessons for research ethics. Qual Res. 6(3):283–99
Download references
Author information
Authors and affiliations.
Department of Public Health, School of Health and Life Sciences, North South University, Dhaka, Bangladesh
Juwel Rana & Segufta Dilshad
Department of Biostatistics and Epidemiology, School of Health and Health Sciences, University of Massachusetts Amherst, Amherst, MA, USA
Department of Research and Innovation, South Asia Institute for Social Transformation (SAIST), Dhaka, Bangladesh
Space and Environment Research Center (SERC), Rajshahi, Bangladesh
Md. Ali Ahsan
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Juwel Rana .
Editor information
Editors and affiliations.
Florida Atlantic University, Boca Raton, FL, USA
Ali Farazmand
Rights and permissions
Reprints and permissions
Copyright information
© 2021 Springer Nature Switzerland AG
About this entry
Cite this entry.
Rana, J., Dilshad, S., Ahsan, M.A. (2021). Ethical Issues in Research. In: Farazmand, A. (eds) Global Encyclopedia of Public Administration, Public Policy, and Governance. Springer, Cham. https://doi.org/10.1007/978-3-319-31816-5_462-1
Download citation
DOI : https://doi.org/10.1007/978-3-319-31816-5_462-1
Received : 01 February 2021
Accepted : 14 February 2021
Published : 05 March 2021
Publisher Name : Springer, Cham
Print ISBN : 978-3-319-31816-5
Online ISBN : 978-3-319-31816-5
eBook Packages : Springer Reference Economics and Finance Reference Module Humanities and Social Sciences Reference Module Business, Economics and Social Sciences
- Publish with us
Policies and ethics
- Find a journal
- Track your research

- LEARNING SKILLS
- Writing a Dissertation or Thesis
Ethical Issues in Research
Search SkillsYouNeed:
Learning Skills:
- A - Z List of Learning Skills
- What is Learning?
- Learning Approaches
- Learning Styles
- 8 Types of Learning Styles
- Understanding Your Preferences to Aid Learning
- Lifelong Learning
- Decisions to Make Before Applying to University
- Top Tips for Surviving Student Life
- Living Online: Education and Learning
- 8 Ways to Embrace Technology-Based Learning Approaches
- Critical Thinking Skills
- Critical Thinking and Fake News
- Understanding and Addressing Conspiracy Theories
- Critical Analysis
- Study Skills
- Exam Skills
- How to Write a Research Proposal
- Dissertation: The Introduction
- Researching and Writing a Literature Review
- Writing your Methodology
- Dissertation: Results and Discussion
- Dissertation: Conclusions and Extras
Writing Your Dissertation or Thesis eBook

Part of the Skills You Need Guide for Students .
- Research Methods
- Teaching, Coaching, Mentoring and Counselling
- Employability Skills for Graduates
Subscribe to our FREE newsletter and start improving your life in just 5 minutes a day.
You'll get our 5 free 'One Minute Life Skills' and our weekly newsletter.
We'll never share your email address and you can unsubscribe at any time.
Ethics are broadly the set of rules, written and unwritten, that govern our expectations of our own and others’ behaviour.
Effectively, they set out how we expect others to behave, and why. While there is broad agreement on some ethical values (for example, that murder is bad), there is also wide variation on how exactly these values should be interpreted in practice.
Research ethics are the set of ethics that govern how scientific and other research is performed at research institutions such as universities, and how it is disseminated.
This page explains more about research ethics, and how you can ensure that your research is compliant.
What are Research Ethics?
When most people think of research ethics, they think about issues that arise when research involves human or animal subjects.
While these issues are indeed a key part of research ethics, there are also wider issues about standards of conduct. These include the importance of publishing findings in a transparent way, not plagiarising others’ work, and not falsifying work.
The Importance of Research Ethics
Research ethics are important for a number of reasons.
- They promote the aims of research, such as expanding knowledge.
- They support the values required for collaborative work, such as mutual respect and fairness. This is essential because scientific research depends on collaboration between researchers and groups.
- They mean that researchers can be held accountable for their actions. Many researchers are supported by public money, and regulations on conflicts of interest, misconduct, and research involving humans or animals are necessary to ensure that money is spent appropriately.
- They ensure that the public can trust research. For people to support and fund research, they have to be confident in it.
- They support important social and moral values, such as the principle of doing no harm to others.
Source: Resnick, D. B. (2015) What is Ethics in Research and Why is it Important?
Codes of Ethics
Government agencies who fund or commission research often publish codes of conduct for researchers, or codes of ethics.
For example, the US National Institutes of Health (NIH) and Food and Drug Administration (FDA) both publish ethical codes. Some ethical codes may have the force of law behind them, while others may simply be advisable.
Be aware that even if you do nothing illegal, doing something unethical may end your research career.
Many or even most ethical codes cover the following areas:
Honesty and Integrity
This means that you need to report your research honestly, and that this applies to your methods (what you did), your data, your results, and whether you have previously published any of it. You should not make up any data, including extrapolating unreasonably from some of your results, or do anything which could be construed as trying to mislead anyone. It is better to undersell than over-exaggerate your findings.
When working with others, you should always keep to any agreements, and act sincerely.
Objectivity
You should aim to avoid bias in any aspect of your research, including design, data analysis, interpretation, and peer review. For example, you should never recommend as a peer reviewer someone you know, or who you have worked with, and you should try to ensure that no groups are inadvertently excluded from your research. This also means that you need to disclose any personal or financial interests that may affect your research.
Carefulness
Take care in carrying out your research to avoid careless mistakes. You should also review your work carefully and critically to ensure that your results are credible. It is also important to keep full records of your research. If you are asked to act as a peer reviewer, you should take the time to do the job effectively and fully.
You should always be prepared to share your data and results, along with any new tools that you have developed, when you publish your findings, as this helps to further knowledge and advance science. You should also be open to criticism and new ideas.
Respect for Intellectual Property
You should never plagiarise, or copy, other people’s work and try to pass it off as your own. You should always ask for permission before using other people’s tools or methods, unpublished data or results. Not doing so is plagiarism. Obviously, you need to respect copyrights and patents, together with other forms of intellectual property, and always acknowledge contributions to your research. If in doubt, acknowledge, to avoid any risk of plagiarism.
Confidentiality
You should respect anything that has been provided in confidence. You should also follow guidelines on protection of sensitive information such as patient records.
Responsible Publication
You should publish to advance to state of research and knowledge, and not just to advance your career. This means, in essence, that you should not publish anything that is not new, or that duplicates someone else’s work.
You should always be aware of laws and regulations that govern your work, and be sure that you conform to them.
Animal Care
If you are using animals in your research, you should always be sure that your experiments are both necessary and well-designed. You should also show respect for the animals you are using, and make sure that they are properly cared for.
Human Subjects Protection
If your research involves people, you should make sure that you reduce any possible harm to the minimum, and maximise the benefits both to participants and other people.
This means, for example, that you should not expose people to more tests than are strictly necessary to fulfil your research aims. You should always respect human rights, including the right to privacy and autonomy. You may need to take particular care with vulnerable groups, which include, but are not limited to, children, older people, and those with learning difficulties.
Source: Resnick, D. B. (2015) What is Ethics in Research and Why is it Important? List adapted from Shamoo A and Resnik D. 2015. Responsible Conduct of Research, 3rd ed. (New York: Oxford University Press).
Further Reading from Skills You Need

The Skills You Need Guide for Students

Develop the skills you need to make the most of your time as a student.
Our eBooks are ideal for students at all stages of education, school, college and university. They are full of easy-to-follow practical information that will help you to learn more effectively and get better grades.
The Role of the Ethics Committee
Most universities have an ethics committee. This is required to scrutinise all research proposals, to ensure that they do not raise any ethical issues. This will generally include research for master’s and undergraduate degrees, although undergraduate research may be covered by a broader research proposal from your supervisor.
There is likely to be a standard form to complete for ethical approval, which will cover who will be involved, how you will recruit your participants, and what steps you will take to ensure that they have provided informed consent.
There is an example form on our page Writing a Research Proposal , which also contains more detail about how to go about preparing a proposal.
The ethics committee’s role is to consider that what you are doing is appropriate and proportionate to your research aims.
If a research proposal raises ethical issues, the committee will ask the researcher to look again at the issue, and consider whether they could do it differently.
For example , if you are proposing to carry out a study on a particular disease, and you want to ask all your participants whether they are married and have any children, the committee may want to know why this is relevant. It may be relevant (for example, if you think the disease may be reduced by living in a family), in which case, you will need to justify this.
The committee may also suggest alternative methods that they think are more suitable for the target group, or additional precautions that you should take.
You cannot start your research until you have been granted ethical approval, which will be granted formally, together with an approval number.
When you publish your research, whether as a thesis or in one or more journal articles, you will need to provide details of the ethical approval, including this number.
If you are unsure how to behave in a particular situation…
…and think you may have an ethical dilemma, then you should always seek advice before you act.
If you are a student, your supervisor should be happy to help and advise you. If necessary, they will be able to advise you about who else to ask.
As a researcher, you should consult more senior colleagues around, either at your own institution or others, who should be happy to help you.
After all, it is in everyone’s interests to promote research ethics, and support the integrity and reputation of research.
Continue to: Designing Research Writing a Methodology
See also: Writing a Literature Review Academic Referencing Sources of Information

How to Address Ethical Issues in Human Subjects Research

Industry Advice Regulatory Affairs
In highly regulated industries such as medical devices, pharmaceuticals, biotechnology, and food safety, companies must constantly balance the demands of bringing new products to market quickly while also testing those products in a safe manner. Many of these products are tested on human subjects, and successful studies can improve human health while helping researchers learn more about diseases, human behavior, and learning patterns.
A mix of national regulations and international declarations offer numerous principles for human subjects research ethics. “The most important thing is to protect the people participating. It goes back to how vulnerable groups have been taken advantage of,” says Mary Dronitsky, a professor in the College of Professional Studies .
This article provides a closer look at three key documents that define the principles of human subjects research ethics—the Nuremberg Code, the Declaration of Helsinki, and the Belmont Report—and offers a set of best practices for regulatory affairs professionals who manage the development, marketing approval, and utilization of highly regulated products.
Download Our Free Guide to Advancing Your Regulatory Affairs Career
Learn how to navigate the discipline and accelerate your regulatory career.
DOWNLOAD NOW
Principles of Human Subjects Research Ethics
National and international organizations have issued declarations on human subjects research ethics in response to medical experiments throughout history that were done on participants who did not consent to being treated, Dronitsky says. These include experiments on prisoners of war in German concentration camps in World War II, the Tuskegee Syphilis Study in the United States from 1932 to 1972, and various vaccine or pain study trials that took advantage of persons with mental disabilities or other underrepresented groups. The three major declarations each consist of several principles of research that act as scientific and moral guidelines for treating people who have agreed to be part of a research study.
Nuremberg Code
Issued by the three judges who presided over the trials of German doctors conducting experiments in World War II, the Nuremberg Code consisted of 10 principles for research trials involving human subjects. First and foremost, the code declares that participant consent is “absolutely essential” and that participants are “at liberty” to bring the experiment to an end at any time.
Additional principles state that experiments must be conducted “for the good of society,” should not cause physical and mental injury to participants, and should not be more risky or dangerous than the problem they are trying to solve.
In a 50th anniversary retrospective, the New England Journal of Medicine noted that the Nuremberg Code has had a “profound” impact on human rights laws and medical ethics even though the code itself has never been adopted as law. For example, its guidelines on informed consent have been the basis for United Nations law and World Health Organization guidelines.
Declaration of Helsinki
Initially adopted by the World Medical Association in 1964 and amended several times since, the Declaration of Helsinki has influenced legislation and regulation worldwide. The document’s principles similarly state that the health of participants must be a physician’s “first consideration.” The document also states that it is the responsibility of healthcare professionals to protect research subjects, even though participants consent to participating.
The Declaration of Helsinki also calls for all research experiments to include a research protocol that indicates how principles of human subjects research ethics are being addressed, along with information about who is funding or otherwise sponsoring the research.
Belmont Report
Written in 1978 in response to the Tuskegee study—in which 400 Black men with syphilis were neither informed of their diagnosis nor treated for the disease for decades—the Belmont Report provides guidance for the treatment of human subjects in research trials in the United States. The report builds on both the Nuremberg Code and the Declaration of Helsinki and consists of three key principles.
- Autonomy : This principle is based on the concept of informed consent , Dronitsky says. “When individuals are asked to participate in a clinical study, they need to be doing it voluntarily and should not face undue pressure. They must also be given sufficient information.”
- Beneficence : The risks of an experiment cannot outweigh the rewards. In addition, the risks and rewards should be clear enough that they can be interpreted by the general public, not just trial participants.
- Justice : The recruitment of volunteers must be impartial, all participants in an experiment must be treated fairly, and participants should not be exploited. “You need or provide full access to all individuals to have the opportunity to be in clinical trials—not just prisoners, those with mental disabilities, or the economically disadvantaged,” Dronitsky says.
Best Practices for Human Subjects Research Ethics
To adhere to these key principles for human subjects research ethics, organizations need to abide by rules established by the U.S. Food and Drug Administration (FDA) and regulatory agencies in any counties where a trial is taking place. A series of simple but critical best practices will help organizations ensure that a trial meets these requirements and can proceed without revisions or other delays.
1. Review consent forms.
Under FDA rules, a clinical trial that requires human subjects must have a consent form that is reviewed and approved by an Ethics Committee or Institutional Review Board (IRB). These rules also apply to physicians working in private practice, who are required to submit research proposals to a hospital, medical school, or local or state government agency. Ultimately, the IRB serves as a “check and balance” for the study, Dronitsky says, ensuring that the needs of the participants and the research teams are aligned.
FDA guidance for IRBs includes specific recommendations for obtaining informed consent, such as conducting informed consent interviews and informing participants of changes to the research study.
The FDA also provides compensation guidelines for trial participants, which the IRB must also review and accept. The agency suggests that payment should be “just and fair” without presenting undue influence that may coerce a participant to provide certain feedback, remain in a study when they would otherwise withdraw, or otherwise interfere with their ability to give voluntary, informed consent.
2. Review recruitment materials.
In the United States, the FDA also requires the IRB to look at the trial’s recruitment materials. “The IRB wants to make sure they are not unduly influencing patients or making promises that the drug will work,” Dronitsky says.
FDA guidance for recruiting human subjects stipulates that advertising material should not include terms such as “new treatment” or “new drug” without indicating that the treatment is experimental. In addition, recruitment material should not promise “free medical treatment” when the standard is not to charge participants to take part in a trial.
The recruitment material that’s reviewed should also include any information that’s given to the office personnel who first speak to potential trial participants, the FDA says. This material should help office personnel address the ways that personal, medical, and financial information are protected as well as take the necessary steps to protect that information.
3. Clearly define participant criteria.
One of the emerging trends in biotechnology is the personalized treatment of rare diseases, made possible through highly targeted treatment plans and therapies that are, in some cases, based on a patient’s genetic and molecular makeup.
These treatments are more effective than generic therapies, and the reduced cost of genetic sequencing has driven down the drug development cost. However, organizations may have difficulty finding participants who meet the very specific criteria for these clinical trials. This roadblock presents safety and ethical issues because organizations must avoid enrolling participants who do not qualify. Not only is the research’s validity in doubt, but the participants may be at a higher risk of an adverse reaction to the treatment, Dronitsky points out.
It’s often not enough to simply recruit patients who have the disease, she continues. Most research study protocols should have a set of inclusion and exclusion criteria that more specifically define and narrow the patient population that’s being recruited. These criteria can include a specific age range, other preexisting conditions, or other medications that someone is taking.
4. Monitor the trial’s progress.
As the experiment continues, investigators must monitor what’s happening to participants. The trial protocols that an IRB approves must include specific requirements for ongoing assessments. Trials that receive funding or other assistance from the National Institutes of Health are also subject to additional oversight, Dronitsky notes.
Assessments may vary depending on the nature of the trial. In a medical device trial, for example, trial organizers need to look at the data that a device generates. If numbers appear to be off, they must further investigate to see if the issue is with the way the device collects and reports data—as with a heart rate monitor in a smartwatch that’s worn incorrectly—or is indicative of a problem that requires medical attention.
In drug trials, meanwhile, the FDA offers guidance for adverse event reporting , which refers to unanticipated problems that arise in patients taking a drug. According to the FDA, certain adverse events must be reported to an IRB, while others do not—but all require investigators to intervene on behalf of a patient’s health and safety.
5. Consider additional expert help.
The sooner that a drug goes to market, the sooner that patients realize clinical benefits and companies enjoy financial benefits for their employees, investors, and shareholders. This can put pressure on investigators to complete a trial quickly, especially if other companies are developing similar drugs or devices.
To facilitate participant recruitment while still adhering to the principles of human subjects research ethics, some organizations opt to hire consultants who specialize in clinical trial recruitment, Dronitsky notes. These individuals can help investigators develop materials to bring in more potential participants—through the use of social media, for example—as well as a more diverse group of participants.
Prepare for a Future in Regulatory Affairs
Professionals in regulatory affairs roles must balance the historical precedents for human subjects research ethics with the reality of rapidly changing regulations in the United States and abroad.
A Master of Science in Regulatory Affairs from Northeastern University helps prepare these professionals to manage the product life cycle in industries such as biotechnology, pharmaceuticals, and medical devices while better understanding the laws and standards that apply to each step of that life cycle.
Download our e-book to learn more about the program and see how it can propel your career in regulatory affairs.

Subscribe below to receive future content from the Graduate Programs Blog.
About shayna joubert, related articles.

4 Ways to Stay Competitive in Regulatory Affairs

Emerging Trends in Regulatory Affairs in 2022

How to Stay Updated on Regulatory Changes
Stay in-demand.
64% of regulatory affairs professionals hold an advanced degree. (RAPS, 2020)
Regulatory Affairs Graduate Programs
Become qualified to manage global regulatory processes and develop cutting-edge products in healthcare and food safety.
Most Popular:
Tips for taking online classes: 8 strategies for success, public health careers: what can you do with an mph, 7 international business careers that are in high demand, edd vs. phd in education: what’s the difference, 7 must-have skills for data analysts, in-demand biotechnology careers shaping our future, the benefits of online learning: 8 advantages of online degrees, how to write a statement of purpose for graduate school, the best of our graduate blog—right to your inbox.
Stay up to date on our latest posts and university events. Plus receive relevant career tips and grad school advice.
By providing us with your email, you agree to the terms of our Privacy Policy and Terms of Service.
Keep Reading:

Should I Go To Grad School: 4 Questions to Consider

Grad School or Work? How to Balance Both

7 Networking Tips for Graduate Students
Research Design Review
A discussion of qualitative & quantitative research design, writing ethics into your qualitative proposal.
Every research proposal for studying human beings must carefully consider the ethical ramifications of engaging individuals for research purposes, and this is particularly true in the relatively intimate, in-depth nature of qualitative research. It is incumbent on qualitative researchers to honestly assure research participants their confidentiality and right to privacy, safety from harm, and right to terminate their voluntary participation at any time with no untoward repercussions from doing so. The proposal should describe the procedures that will be taken to implement these assurances, including gaining informed consent, gaining approval from the relevant Institutional Review Board, and anonymizing participants’ names, places mentioned, and other potentially identifying information.
Special consideration should be given in the proposal to ethical matters when the proposed research (a) pertains to vulnerable populations such as children or the elderly; (b) concerns a marginalized segment of the population such as people with disabilities, same-sex couples, or the economically disadvantaged; (c) involves covert observation that will be conducted in association with an ethnographic study; or (d) is a narrative study in which the researcher may withhold the full true intent of the research in order not to stifle or bias participants’ telling of their stories.
Furthermore, the researcher should pay particular attention to ethical considerations when writing a proposal for a focus group study. The focus group method (regardless of mode) brings together (typically) a number of strangers who are often asked to offer their candid thoughts on personal and sensitive topics. For this reason (and other reasons, e.g., the moderator may be sharing confidential information with the participants), it is important to gain a signed consent form from all participants; however, the reality is that there is no way the researcher can totally guarantee confidentiality. These and other associated ethical considerations should be discussed in the Design section of the focus group proposal.
Share this:
- Click to share on Reddit (Opens in new window)
- Click to share on Twitter (Opens in new window)
- Click to share on LinkedIn (Opens in new window)
- Click to share on Facebook (Opens in new window)
- Click to share on Tumblr (Opens in new window)
- Click to email a link to a friend (Opens in new window)
- Click to print (Opens in new window)
- Pingback: The TQF Qualitative Research Proposal: Limitations | Research Design Review
- Pingback: The TQF Qualitative Research Proposal: The Research Team | Research Design Review
- Pingback: The Total Quality Framework Proposal: Design Section — Credibility | Research Design Review
Great article! Ethical considerations become even more significant as we incorporate more and more technology.
- Pingback: Writing Ethics Into Your Qualitative Proposal | Managementpublic
Leave a comment Cancel reply
This site uses Akismet to reduce spam. Learn how your comment data is processed .

- Already have a WordPress.com account? Log in now.
- Subscribe Subscribed
- Copy shortlink
- Report this content
- View post in Reader
- Manage subscriptions
- Collapse this bar

Ethical Considerations
Ethical Considerations can be specified as one of the most important parts of the research. Dissertations may even be doomed to failure if this part is missing.
According to Bryman and Bell (2007) [1] the following ten points represent the most important principles related to ethical considerations in dissertations:
- Research participants should not be subjected to harm in any ways whatsoever.
- Respect for the dignity of research participants should be prioritised.
- Full consent should be obtained from the participants prior to the study.
- The protection of the privacy of research participants has to be ensured.
- Adequate level of confidentiality of the research data should be ensured.
- Anonymity of individuals and organisations participating in the research has to be ensured.
- Any deception or exaggeration about the aims and objectives of the research must be avoided.
- Affiliations in any forms, sources of funding, as well as any possible conflicts of interests have to be declared.
- Any type of communication in relation to the research should be done with honesty and transparency.
- Any type of misleading information, as well as representation of primary data findings in a biased way must be avoided.
In order to address ethical considerations aspect of your dissertation in an effective manner, you will need to expand discussions of each of the following points to at least one paragraph:
1. Voluntary participation of respondents in the research is important. Moreover, participants have rights to withdraw from the study at any stage if they wish to do so.
2. Respondents should participate on the basis of informed consent. The principle of informed consent involves researchers providing sufficient information and assurances about taking part to allow individuals to understand the implications of participation and to reach a fully informed, considered and freely given decision about whether or not to do so, without the exercise of any pressure or coercion. [2]
3. The use of offensive, discriminatory, or other unacceptable language needs to be avoided in the formulation of Questionnaire/Interview/Focus group questions.
4. Privacy and anonymity or respondents is of a paramount importance.
5. Acknowledgement of works of other authors used in any part of the dissertation with the use of Harvard/APA/Vancouver referencing system according to the Dissertation Handbook
6. Maintenance of the highest level of objectivity in discussions and analyses throughout the research
7. Adherence to Data Protection Act (1998) if you are studying in the UK
In studies that do not involve primary data collection, on the other hand, ethical issues are going to be limited to the points d) and e) above.
Most universities have their own Code of Ethical Practice. It is critically important for you to thoroughly adhere to this code in every aspect of your research and declare your adherence in ethical considerations part of your dissertation.
My e-book, The Ultimate Guide to Writing a Dissertation in Business Studies: a step by step assistance offers practical assistance to complete a dissertation with minimum or no stress. The e-book covers all stages of writing a dissertation starting from the selection to the research area to submitting the completed version of the work within the deadline. John Dudovskiy

[1] Bryman, A. & Bell, E. (2007) “Business Research Methods”, 2nd edition. Oxford University Press.
[2] Saunders, M., Lewis, P. & Thornhill, A. (2012) “Research Methods for Business Students” 6th edition, Pearson Education Limited.

Dissertations - Skills Guide
- Where to start
- Research Proposal
Ethics Form
- Primary Research
- Literature Review
- Methodology
- Downloadable Resources
- Further Reading
What is it?
An ethics form is a document that prompts you to provide information about your research to ensure you are meeting set standards. Readers usually expect to see ethics in a research proposal, or mentioned in your writing, even if there doesn't appear to be any problematic ethical issues to be addressed.
Why do I need to do it?
When someone embarks on a piece of research there is a chance of doing harm, even if harm isn't intended. Setting ethical guidelines ensures there are set standards for conducting research to ensure the research will not harm people physically or emotionally.
How do I do it?
You can find more information about completing your ethics form from the research ethics page of the university website ( click here ).
Ethics Further Reading
- << Previous: Research Proposal
- Next: Primary Research >>
- Last Updated: Oct 18, 2023 9:32 AM
- URL: https://libguides.derby.ac.uk/c.php?g=690330

Library Guides
Dissertations 4: methodology: ethics.
- Introduction & Philosophy
- Methodology
Research Ethics
In the research context, ethics can be defined as "the standards of behaviour that guide your conduct in relation to the rights of those who become the subject of your work, or are affected by it" (Saunders, Lewis and Thornhill 2015, p239).
The University itself is guided by the fundamental principle that research involving humans and /or animals and/or the environment should involve no more than minimal risk of harm to physical and psychological wellbeing.
Thus, ethics relates to many aspects of your research, including the conduct towards:
The participants of your primary research (experiments, interviews etc). You will need to explain that participation is voluntary, and they have the right to withdraw at any time. You will need the participants' informed consent. You will need to avoid harming the participants, physically as well as mentally. You will need to respect the participants’ privacy and offer the right to anonymity. You will need to manage their personal data confidentially, also according to legislation such as the Data Protection Act 2018. You will need to be truthful and accurate when using the information provided by the participants.
The authors you have used as secondary sources. You will need to acknowledge their work and avoid plagiarism by doing the proper citing and referencing.
The readers of your research. You will need to exercise the utmost integrity, honesty, accuracy and objectivity in the writing of your work.
The researcher . You will need to ensure that the research will be safe for you to undertake.
Your research may entail some risk, but risk has to be analysed and minimised through risk assessment. Depending on the type of your research, your research proposal may need to be approved by an Ethics Committee, which will assess your research proposal in light of the elements mentioned above. Again, you are advised to use a research methods book for further guidance.
Research Ethics Online Course
Introduction to Research Ethics: Working with People
Find out how to conduct ethical research when working with people by studying this online course for university students. Course developed by the University of Leeds.

- << Previous: Methods
- Next: Methodology >>
- Last Updated: Sep 14, 2022 12:58 PM
- URL: https://libguides.westminster.ac.uk/methodology-for-dissertations
CONNECT WITH US

Ethics statement examples - ESRC
Introduction.
Proposals submitted to the ESRC must provide a full ethics statement that confirms that proper consideration has been given to any ethics issues raised. All ESRC-funded grants must be approved by at least a light-touch ethics review.
The ESRC does not require a favourable ethics opinion to be secured prior to submission of a research proposal. However, a proposal must state what the applicant considers to be the possible ethics implications throughout the research project lifecycle, what measures will be taken for ongoing consideration of ethics issues, what review will be required for their proposed research and how and when it will be obtained.
Risk and benefit to researchers, participants and others (for example, potentially stigmatised or marginalised groups) as a result of the research and the potential impact, knowledge exchange, dissemination activity and future re-use of the data should also be considered as part of the ethical statement.
If an ethics review is required at a later stage in the project, this should be discussed and funding arrangements agreed in advance with the ESRC. At a minimum we expect that ethics review will be undertaken prior to the stage in the project that the actual research is carried out.
During peer review, reviewers and assessors will be asked to consider the ethical statement in the proposal. If they disagree with the proposed approach to ethics issues, or the statement does not adequately address these issues, this could lead to the rejection of a proposal, or the award of a conditional grant to ensure the necessary ethical considerations and ethical review are undertaken.
Last updated: 28 January 2022
This is the website for UKRI: our seven research councils, Research England and Innovate UK. Let us know if you have feedback or would like to help improve our online products and services .
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- Indian J Anaesth
- v.60(9); 2016 Sep
How to write a research proposal?
Department of Anaesthesiology, Bangalore Medical College and Research Institute, Bengaluru, Karnataka, India
Devika Rani Duggappa
Writing the proposal of a research work in the present era is a challenging task due to the constantly evolving trends in the qualitative research design and the need to incorporate medical advances into the methodology. The proposal is a detailed plan or ‘blueprint’ for the intended study, and once it is completed, the research project should flow smoothly. Even today, many of the proposals at post-graduate evaluation committees and application proposals for funding are substandard. A search was conducted with keywords such as research proposal, writing proposal and qualitative using search engines, namely, PubMed and Google Scholar, and an attempt has been made to provide broad guidelines for writing a scientifically appropriate research proposal.
INTRODUCTION
A clean, well-thought-out proposal forms the backbone for the research itself and hence becomes the most important step in the process of conduct of research.[ 1 ] The objective of preparing a research proposal would be to obtain approvals from various committees including ethics committee [details under ‘Research methodology II’ section [ Table 1 ] in this issue of IJA) and to request for grants. However, there are very few universally accepted guidelines for preparation of a good quality research proposal. A search was performed with keywords such as research proposal, funding, qualitative and writing proposals using search engines, namely, PubMed, Google Scholar and Scopus.
Five ‘C’s while writing a literature review

BASIC REQUIREMENTS OF A RESEARCH PROPOSAL
A proposal needs to show how your work fits into what is already known about the topic and what new paradigm will it add to the literature, while specifying the question that the research will answer, establishing its significance, and the implications of the answer.[ 2 ] The proposal must be capable of convincing the evaluation committee about the credibility, achievability, practicality and reproducibility (repeatability) of the research design.[ 3 ] Four categories of audience with different expectations may be present in the evaluation committees, namely academic colleagues, policy-makers, practitioners and lay audiences who evaluate the research proposal. Tips for preparation of a good research proposal include; ‘be practical, be persuasive, make broader links, aim for crystal clarity and plan before you write’. A researcher must be balanced, with a realistic understanding of what can be achieved. Being persuasive implies that researcher must be able to convince other researchers, research funding agencies, educational institutions and supervisors that the research is worth getting approval. The aim of the researcher should be clearly stated in simple language that describes the research in a way that non-specialists can comprehend, without use of jargons. The proposal must not only demonstrate that it is based on an intelligent understanding of the existing literature but also show that the writer has thought about the time needed to conduct each stage of the research.[ 4 , 5 ]
CONTENTS OF A RESEARCH PROPOSAL
The contents or formats of a research proposal vary depending on the requirements of evaluation committee and are generally provided by the evaluation committee or the institution.
In general, a cover page should contain the (i) title of the proposal, (ii) name and affiliation of the researcher (principal investigator) and co-investigators, (iii) institutional affiliation (degree of the investigator and the name of institution where the study will be performed), details of contact such as phone numbers, E-mail id's and lines for signatures of investigators.
The main contents of the proposal may be presented under the following headings: (i) introduction, (ii) review of literature, (iii) aims and objectives, (iv) research design and methods, (v) ethical considerations, (vi) budget, (vii) appendices and (viii) citations.[ 4 ]
Introduction
It is also sometimes termed as ‘need for study’ or ‘abstract’. Introduction is an initial pitch of an idea; it sets the scene and puts the research in context.[ 6 ] The introduction should be designed to create interest in the reader about the topic and proposal. It should convey to the reader, what you want to do, what necessitates the study and your passion for the topic.[ 7 ] Some questions that can be used to assess the significance of the study are: (i) Who has an interest in the domain of inquiry? (ii) What do we already know about the topic? (iii) What has not been answered adequately in previous research and practice? (iv) How will this research add to knowledge, practice and policy in this area? Some of the evaluation committees, expect the last two questions, elaborated under a separate heading of ‘background and significance’.[ 8 ] Introduction should also contain the hypothesis behind the research design. If hypothesis cannot be constructed, the line of inquiry to be used in the research must be indicated.
Review of literature
It refers to all sources of scientific evidence pertaining to the topic in interest. In the present era of digitalisation and easy accessibility, there is an enormous amount of relevant data available, making it a challenge for the researcher to include all of it in his/her review.[ 9 ] It is crucial to structure this section intelligently so that the reader can grasp the argument related to your study in relation to that of other researchers, while still demonstrating to your readers that your work is original and innovative. It is preferable to summarise each article in a paragraph, highlighting the details pertinent to the topic of interest. The progression of review can move from the more general to the more focused studies, or a historical progression can be used to develop the story, without making it exhaustive.[ 1 ] Literature should include supporting data, disagreements and controversies. Five ‘C's may be kept in mind while writing a literature review[ 10 ] [ Table 1 ].
Aims and objectives
The research purpose (or goal or aim) gives a broad indication of what the researcher wishes to achieve in the research. The hypothesis to be tested can be the aim of the study. The objectives related to parameters or tools used to achieve the aim are generally categorised as primary and secondary objectives.
Research design and method
The objective here is to convince the reader that the overall research design and methods of analysis will correctly address the research problem and to impress upon the reader that the methodology/sources chosen are appropriate for the specific topic. It should be unmistakably tied to the specific aims of your study.
In this section, the methods and sources used to conduct the research must be discussed, including specific references to sites, databases, key texts or authors that will be indispensable to the project. There should be specific mention about the methodological approaches to be undertaken to gather information, about the techniques to be used to analyse it and about the tests of external validity to which researcher is committed.[ 10 , 11 ]
The components of this section include the following:[ 4 ]
Population and sample
Population refers to all the elements (individuals, objects or substances) that meet certain criteria for inclusion in a given universe,[ 12 ] and sample refers to subset of population which meets the inclusion criteria for enrolment into the study. The inclusion and exclusion criteria should be clearly defined. The details pertaining to sample size are discussed in the article “Sample size calculation: Basic priniciples” published in this issue of IJA.
Data collection
The researcher is expected to give a detailed account of the methodology adopted for collection of data, which include the time frame required for the research. The methodology should be tested for its validity and ensure that, in pursuit of achieving the results, the participant's life is not jeopardised. The author should anticipate and acknowledge any potential barrier and pitfall in carrying out the research design and explain plans to address them, thereby avoiding lacunae due to incomplete data collection. If the researcher is planning to acquire data through interviews or questionnaires, copy of the questions used for the same should be attached as an annexure with the proposal.
Rigor (soundness of the research)
This addresses the strength of the research with respect to its neutrality, consistency and applicability. Rigor must be reflected throughout the proposal.
It refers to the robustness of a research method against bias. The author should convey the measures taken to avoid bias, viz. blinding and randomisation, in an elaborate way, thus ensuring that the result obtained from the adopted method is purely as chance and not influenced by other confounding variables.
Consistency
Consistency considers whether the findings will be consistent if the inquiry was replicated with the same participants and in a similar context. This can be achieved by adopting standard and universally accepted methods and scales.
Applicability
Applicability refers to the degree to which the findings can be applied to different contexts and groups.[ 13 ]
Data analysis
This section deals with the reduction and reconstruction of data and its analysis including sample size calculation. The researcher is expected to explain the steps adopted for coding and sorting the data obtained. Various tests to be used to analyse the data for its robustness, significance should be clearly stated. Author should also mention the names of statistician and suitable software which will be used in due course of data analysis and their contribution to data analysis and sample calculation.[ 9 ]
Ethical considerations
Medical research introduces special moral and ethical problems that are not usually encountered by other researchers during data collection, and hence, the researcher should take special care in ensuring that ethical standards are met. Ethical considerations refer to the protection of the participants' rights (right to self-determination, right to privacy, right to autonomy and confidentiality, right to fair treatment and right to protection from discomfort and harm), obtaining informed consent and the institutional review process (ethical approval). The researcher needs to provide adequate information on each of these aspects.
Informed consent needs to be obtained from the participants (details discussed in further chapters), as well as the research site and the relevant authorities.
When the researcher prepares a research budget, he/she should predict and cost all aspects of the research and then add an additional allowance for unpredictable disasters, delays and rising costs. All items in the budget should be justified.
Appendices are documents that support the proposal and application. The appendices will be specific for each proposal but documents that are usually required include informed consent form, supporting documents, questionnaires, measurement tools and patient information of the study in layman's language.
As with any scholarly research paper, you must cite the sources you used in composing your proposal. Although the words ‘references and bibliography’ are different, they are used interchangeably. It refers to all references cited in the research proposal.
Successful, qualitative research proposals should communicate the researcher's knowledge of the field and method and convey the emergent nature of the qualitative design. The proposal should follow a discernible logic from the introduction to presentation of the appendices.
Financial support and sponsorship
Conflicts of interest.
There are no conflicts of interest.

IMAGES
VIDEO
COMMENTS
Research ethics are a set of principles that guide your research designs and practices in both quantitative and qualitative research. In this article, you will learn about the types and examples of ethical considerations in research, such as informed consent, confidentiality, and avoiding plagiarism. You will also find out how to apply ethical principles to your own research projects with ...
Principles that come to mind might include autonomy, respect, dignity, privacy, informed consent and confidentiality. You may also have identified principles such as competence, integrity, wellbeing, justice and non-discrimination. Key ethical issues that you will address as an insider researcher include: Gaining trust.
How to Write Ethical Considerations. When writing about research involving human subjects or animals, it is essential to include ethical considerations to ensure that the study is conducted in a manner that is morally responsible and in accordance with professional standards. Here are some steps to help you write ethical considerations:
Writing the Research Proposal ... Anticipated ethical issues, and approaches you will use to address these issues. Preliminary pilot findings (if available) Expected impact and significance of study This is the what of the proposal. What do you hope to accomplish, at what level? Will it potentially make an impact on individuals, groups ...
Underpayment is a common ethical issue that shouldn't be overlooked. Properly rewarding participants' time is critical for boosting engagement and obtaining high-quality data. While Prolific requires a minimum payment of £6.00 / $8.00 per hour, there are other factors you need to consider when deciding on a fair payment.
Here are some key reasons why research ethics matter: Research ethics protect the rights and well-being of participants, uphold the integrity of research findings, and contribute to the positive impact of research on individuals and society. Let us look into some of the major ethical considerations in research design. Ethical Issues in Research
These ethical issues should be discussed at the proposal stage, and the participants/guardians should be informed about the decision to report to the police while taking the consent. This will potentially affect the response rate; but this is also the responsibility of the researcher to protect the participants and their families.
In the sections to follow, we map out the various ethical dimensions of designing a research project step by step: addressing the fundamental question of why and for whom we do research (Sect. 10.2); an exploration of the ethical considerations of the research design itself, including the recruitment of study participants (Sects. 10.3 and 10.4 ...
Research is a critical aspect of any design journey, but ensuring that the research is accurate, unbiased, and ethical is just as important.. Whether you are conducting a survey, running focus groups, doing field research, or holding interviews, the chances are participants will be a part of the process.. Taking ethical considerations into account and following all obligations are essential ...
Definition. Ethics is a set of standards, a code, or value system, worked out from human reason and experience, by which free human actions are determined as ultimately right or wrong, good, or evil. If acting agrees with these standards, it is ethical, otherwise unethical. Scientific research refers to a persistent exercise towards producing ...
There is an example form on our page Writing a Research Proposal, which also contains more detail about how to go about preparing a proposal. The ethics committee's role is to consider that what you are doing is appropriate and proportionate to your research aims. If a research proposal raises ethical issues, the committee will ask the ...
We discuss two issues related to ethics in scientific writing: Plagiarism and authorship. Plagiarism, the most common form of scientific misconduct, is defined as the appropriation of another person's ideas, processes, results or words without giving appropriate credit. While plagiarism is often intentional, it may be unintentional due to ...
to engage in ethical practices and to anticipate the ethical issues prior to the study that will likely arise. This chapter provides guidance for the overall structure of proposals or projects, writing practices that make projects easy to read, and ethical issues that need to be anticipated in research studies.
1. Review consent forms. Under FDA rules, a clinical trial that requires human subjects must have a consent form that is reviewed and approved by an Ethics Committee or Institutional Review Board (IRB). These rules also apply to physicians working in private practice, who are required to submit research proposals to a hospital, medical school ...
Writing Ethics Into Your Qualitative Proposal. A qualitative research proposal is comprised of many pieces and parts that are necessary to convey the researcher's justification for conducting the research, how the research will be conducted (including the strengths and limitations of the pr oposed approach), as well as what the sponsor of the ...
What are research ethics? Research ethics are the moral principles that govern how researchers should carry out their work. These principles are used to shape research regulations agreed by groups such as university governing bodies, communities or governments. All researchers should follow any regulations that apply to their work.
In order to address ethical considerations aspect of your dissertation in an effective manner, you will need to expand discussions of each of the following points to at least one paragraph: 1. Voluntary participation of respondents in the research is important. Moreover, participants have rights to withdraw from the study at any stage if they ...
An ethics form is a document that prompts you to provide information about your research to ensure you are meeting set standards. Readers usually expect to see ethics in a research proposal, or mentioned in your writing, even if there doesn't appear to be any problematic ethical issues to be addressed. Why do I need to do it? When someone ...
The different ratings indicate differences in the groups' perspectives on ethical principles, which may affect focal congruence on ethical issues in the proposal. Communication of these findings should help close gaps between REC and researcher perceptions. Further study should investigate how RECs and researchers perceive equivocal ethics terms.
Research Ethics. In the research context, ethics can be defined as "the standards of behaviour that guide your conduct in relation to the rights of those who become the subject of your work, or are affected by it" (Saunders, Lewis and Thornhill 2015, p239). The University itself is guided by the fundamental principle that research involving ...
Introduction. Proposals submitted to the ESRC must provide a full ethics statement that confirms that proper consideration has been given to any ethics issues raised. All ESRC-funded grants must be approved by at least a light-touch ethics review. The ESRC does not require a favourable ethics opinion to be secured prior to submission of a ...
Introduction. Research includes a set of activities in which researchers use various structured methods to contribute to the development of knowledge, whether this knowledge is theoretical, fundamental, or applied (Drolet & Ruest, accepted).University research is carried out in a highly competitive environment that is characterized by ever-increasing demands (i.e., on time, productivity ...
A proposal needs to show how your work fits into what is already known about the topic and what new paradigm will it add to the literature, while specifying the question that the research will answer, establishing its significance, and the implications of the answer. [ 2] The proposal must be capable of convincing the evaluation committee about ...